Uncategorized
TGA approves trial for first-in-human multiple myeloma therapy
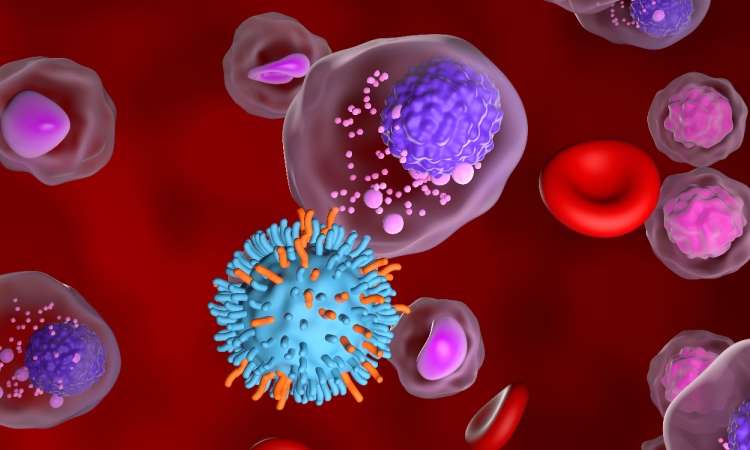

Australia’s Therapeutic Goods Administration (TGA) has approved a first-in-human trial of a T-cell therapy in multiple myeloma.
The therapy, developed by HaemaLogiX in collaboration with the Peter MacCallum Cancer Centre (Peter Mac), is a KMA.CAR T-cell immunotherapy. The novel autologous CAR-T cell therapy targets Kappa Myeloma Antigen (KMA), a receptor found only on the surface of myeloma cells and not on healthy immune cells.
This tumour-specific targeting means that, unlike currently approved BCMA-directed CAR-T therapies, KMCAR T-cell is not expected to damage healthy immune cells, potentially offering patients an effective treatment without affecting their natural ability to ward off infection.
KMCAR T-cell therapy, developed with HaemaLogiX’s KappaMab antibody technology, harnesses the patient’s own T cells and genetically modifies them to recognise and destroy cancerous plasma cells that express KMA.
Figures show multiple myeloma is the second most common blood cancer worldwide, with estimations from the World Health Organization (WHO) and Global Cancer Observatory (GLOBOCAN) suggesting approximately 188,000 new cases of multiple myeloma are diagnosed each year globally. Data shows 42% of patients die from the disease within five years of diagnosis.
“Peter Mac’s Centre of Excellence in Cellular Immunotherapy is world-class and has been instrumental in bringing CAR-T therapies from the laboratory to patients,” said Dr Rosanne Dunn, Chief Scientific Officer and Founder of HaemaLogiX.
“We are incredibly fortunate to have such a partner to translate our KMA targeting technology into the CAR-T modality.
“For patients with multiple myeloma who have failed or relapsed on their current treatment, it is critical that we explore novel therapeutic targets. The KMA target is unique – it is expressed on malignant plasma cells but spares normal immune cells.
“Through the CAR-T approach, we have an opportunity to harness this tumour specificity to potentially deliver meaningful benefits for patients without harming their immune system.”
The post TGA approves trial for first-in-human multiple myeloma therapy appeared first on Drug Discovery World (DDW).
Uncategorized
Sanofi Bispecific Scores in Asthma, Rhinosinusitis, but Eczema Bet Doesn’t Pay Off
Sanofi Bispecific Scores in Asthma, Rhinosinusitis, but Eczema Bet Doesn’t Pay Off
Sanofi has faced questions about the potential of lunsekimig in eczema, with executives calling the clinical trial a “measured risk.” Read More
Uncategorized
STAT+: Gilead to buy cancer biotech Tubulis for more than $3 billion
In a move that will grow its oncology pipeline, Gilead Sciences is spending more than $3 billion to buy the German biotech Tubulis, the companies said Tuesday.
The deal includes an upfront payment of $3.15 billion and up to $1.85 billion more if certain milestones are reached. It also highlights both the therapeutic and commercial promise of antibody-drug conjugates, the next-generation chemotherapy treatments that the privately held Tubulis is developing.
Gilead’s latest move comes just months after it said it would acquire Arcellx in a deal worth $7.8 billion. The two companies had already been working together on a multiple myeloma CAR-T therapy that could be approved later this year. Gilead last month also announced that it was buying Ouro Therapeutics, which is focused on drugs for autoimmune disease, for up to $2.18 billion.

In a move that will grow its oncology pipeline, Gilead Sciences is spending more than $3 billion to buy the German biotech Tubulis, the companies said Tuesday.
The deal includes an upfront payment of $3.15 billion and up to $1.85 billion more if certain milestones are reached. It also highlights both the therapeutic and commercial promise of antibody-drug conjugates, the next-generation chemotherapy treatments that the privately held Tubulis is developing.
Gilead’s latest move comes just months after it said it would acquire Arcellx in a deal worth $7.8 billion. The two companies had already been working together on a multiple myeloma CAR-T therapy that could be approved later this year. Gilead last month also announced that it was buying Ouro Therapeutics, which is focused on drugs for autoimmune disease, for up to $2.18 billion.
Uncategorized
Gilead continues dealmaking streak with $3.15B Tubulis buy for ADCs
In its third acquisition of 2026, Gilead is spending $3.15 billion upfront to snag a next-generation antibody-drug conjugate platform from German startup Tubulis.
The California biopharma could pay out another $1.85 billion down the road …
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors




