Uncategorized
Study supports Treg enhancement as dementia treatment
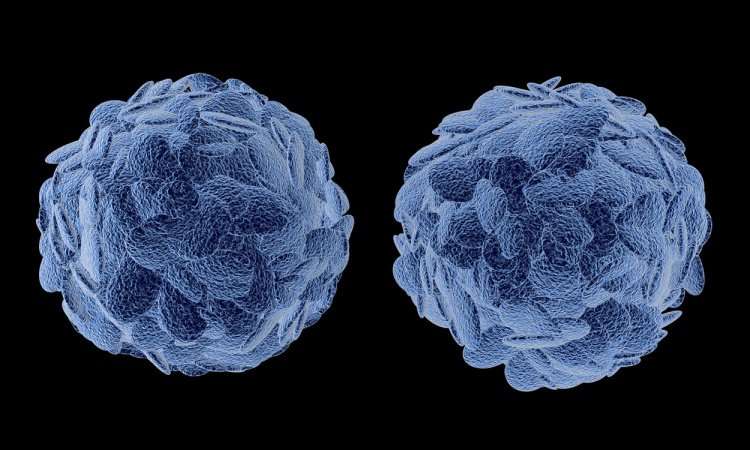

A research study led by Dr Alireza Faridar and Dr Stanley Appel at the Houston Methodist Neurological Institute has demonstrated the involvement of the peripheral immune system in the neuroinflammatory profile of frontotemporal dementia (FTD).
Dr Fred Grossman, Chief Medical Officer at sponsors Coya, said: “We believe these biomarker data coupled with the clinical and lab results of the clinical study in FTD provide additional evidence supporting the development of biologic combination therapies that enhance the function of regulatory T cells (Tregs) and target inflammation in FTD, ALS and other neurodegenerative diseases of high unmet need.”
Coya’s investigational product candidate pipeline aims to restore the anti-inflammatory and immunomodulatory functions of Tregs. Its therapeutic platforms include Treg-enhancing biologics, Treg-derived exosomes, and autologous Treg cell therapy.
Blood samples were obtained from 27 patients clinically diagnosed with FTD and 25 age-matched healthy individuals as a control group. Comprehensive analyses revealed significantly lower regulatory Treg immunomodulatory suppressive function in FTD patients compared to healthy controls. Transcriptomic profiling of monocytes showed different degrees of dysregulation of immune-related genes in samples from FTD patients.
Consistent with the reduced anti-inflammatory function observed in Tregs, proteomic analysis of plasma inflammatory mediators showed a significant increase in the pro-inflammatory cytokine TNFα (tumour necrosis factor-alpha) and the chemokines CXCL10, CCL3, CCL19, CSF1 and CXCL12 in FTD patients compared to healthy controls.
The immunomodulatory function of Tregs
The company believes the results of this study demonstrate that there is a dysregulation of inflammation-related gene expression in peripheral monocytes and an increase of plasma inflammatory chemokines and cytokines in FTD. In addition, the data provide evidence of compromised immunomodulatory function of Tregs.
The company argues that this study further supports the previously reported findings of an academic clinical study of low-dose interleukin 2 (LD IL-2) and CTLA-4 Ig in patients with FTD. The subcutaneous administration of LD IL-2 and CTLA-4 Ig significantly increased the number and function of Tregs as early as two weeks after initiation of treatment and remained elevated throughout the study. Study patients did not show clinical cognitive decline as measured by the validated tools MoCA (Montreal Cognitive Assessment) and CDR-FTLD (Clinical Dementia Rating-Frontotemporal Lobar Degeneration module) over the 22-week treatment period.
The post Study supports Treg enhancement as dementia treatment appeared first on Drug Discovery World (DDW).
Uncategorized
Gilead Swallows Another Partner, Paying up to $5B for ADC Specialist Tubulis
The acquisition of Tubulis GmbH—Gilead Sciences’ latest of the year after buying Arcells and Ouro Medicines—brings into the fold a novel ovarian cancer candidate that has demonstrated promising mid-stage data.
Uncategorized
STAT+: Pharmalittle: We’re reading about FDA backing domestic production, another Gilead deal, and more
Rise and shine, everyone, another busy day is on the way. And it is getting off to a good start here on the Pharmalot campus, where clear blue skies and comfortable breezes are greeting us. Who could ask for anything more? Actually, we could — it is time to reheat the kettle for another cuppa stimulation. Our choice today is ginger peach. And here is a helpful tip — a teaspoon of honey enhances the flavors splendidly. Of course, you are invited to join us. For the full experience, we are now hawking replicas — take a look. Meanwhile, here are a few tidbits to help you along. As always, do keep in touch. We appreciate feedback, criticism, and tips. …
The U.S. Food and Drug Administration used the president’s budget to propose policies aimed at encouraging domestic development and manufacturing of drugs, STAT notes. FDA Commissioner Marty Makary has said the agency needs “giant, big ideas” to counter China’s dominance in early-stage clinical development of drugs. Among the FDA’s ideas are proposals to make it easier to run early-stage trials in the U.S. and to hand an advantage to U.S.-based generics manufacturers. The Trump administration has been using a variety of policy levers to try and bring drug manufacturing to the U.S. One of the legislative proposals in the FDA’s budget justification would let domestic manufacturers of generic drugs challenge brand drug patents a month before foreign companies, a major advantage in an intensely competitive process.
Two more drugmakers, AbbVie and Genentech, will officially start selling their medicines on the TrumpRx website, CBS News tells us. Abbvie, which struck a deal with the Trump administration in January to cut the cost of certain medicines, will sell Humira, a popular medication used to treat rheumatoid arthritis, Crohn’s disease, and ulcerative colitis, on the site at an 86% discount. The prescription prices on the site, however, are only available to patients who are uninsured, or whose insurance does not cover it, and who must pay the full list price out of pocket. Those with insurance coverage generally pay lower prices already. TrumpRx now sells over 61 drugs at a lower price, up from about 40 when the website went live in February.

Rise and shine, everyone, another busy day is on the way. And it is getting off to a good start here on the Pharmalot campus, where clear blue skies and comfortable breezes are greeting us. Who could ask for anything more? Actually, we could — it is time to reheat the kettle for another cuppa stimulation. Our choice today is ginger peach. And here is a helpful tip — a teaspoon of honey enhances the flavors splendidly. Of course, you are invited to join us. For the full experience, we are now hawking replicas — take a look. Meanwhile, here are a few tidbits to help you along. As always, do keep in touch. We appreciate feedback, criticism, and tips. …
The U.S. Food and Drug Administration used the president’s budget to propose policies aimed at encouraging domestic development and manufacturing of drugs, STAT notes. FDA Commissioner Marty Makary has said the agency needs “giant, big ideas” to counter China’s dominance in early-stage clinical development of drugs. Among the FDA’s ideas are proposals to make it easier to run early-stage trials in the U.S. and to hand an advantage to U.S.-based generics manufacturers. The Trump administration has been using a variety of policy levers to try and bring drug manufacturing to the U.S. One of the legislative proposals in the FDA’s budget justification would let domestic manufacturers of generic drugs challenge brand drug patents a month before foreign companies, a major advantage in an intensely competitive process.
Two more drugmakers, AbbVie and Genentech, will officially start selling their medicines on the TrumpRx website, CBS News tells us. Abbvie, which struck a deal with the Trump administration in January to cut the cost of certain medicines, will sell Humira, a popular medication used to treat rheumatoid arthritis, Crohn’s disease, and ulcerative colitis, on the site at an 86% discount. The prescription prices on the site, however, are only available to patients who are uninsured, or whose insurance does not cover it, and who must pay the full list price out of pocket. Those with insurance coverage generally pay lower prices already. TrumpRx now sells over 61 drugs at a lower price, up from about 40 when the website went live in February.
Uncategorized
FDA Seeks Expanded Authority To Regulate Postapproval Manufacturing Changes
FDA Seeks Expanded Authority To Regulate Postapproval Manufacturing Changes
Aiming to protect patients, the FDA sent lawmakers a wish list of legislative proposals intended to clarify and expand its oversight of updates to approved drug production processes. Read More
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors




