Uncategorized
STAT+: Zap in a cap: How one neurotech startup is using a hat to treat depression
Wake up. Brush your teeth. Wash your face.
And put on your lifesaving baseball hat.
That’s right. If you have treatment-resistant depression, this could be the regular morning routine in your future. The hat would activate a blueberry-sized device implanted in your skull that sends a pulse of electricity into your brain.
This is Jacob Robinson’s vision — and it got closer to reality on Friday after the Food and Drug Administration approved a request from Robinson’s startup, Motif Neurotech, to start an initial feasibility trial to test the efficacy of their device in treating depression that hasn’t responded to other treatments. Scientists have been zapping brains to alleviate depression for decades through a method called transcranial magnetic stimulation, or TMS. Motif wants to do the same thing, but with a twist.

Wake up. Brush your teeth. Wash your face.
And put on your lifesaving baseball hat.
That’s right. If you have treatment-resistant depression, this could be the regular morning routine in your future. The hat would activate a blueberry-sized device implanted in your skull that sends a pulse of electricity into your brain.
This is Jacob Robinson’s vision — and it got closer to reality on Friday after the Food and Drug Administration approved a request from Robinson’s startup, Motif Neurotech, to start an initial feasibility trial to test the efficacy of their device in treating depression that hasn’t responded to other treatments. Scientists have been zapping brains to alleviate depression for decades through a method called transcranial magnetic stimulation, or TMS. Motif wants to do the same thing, but with a twist.
Uncategorized
STAT+: Capricor Therapeutics accuses Nippon Shinyaku of slow-walking plans on Duchenne drug
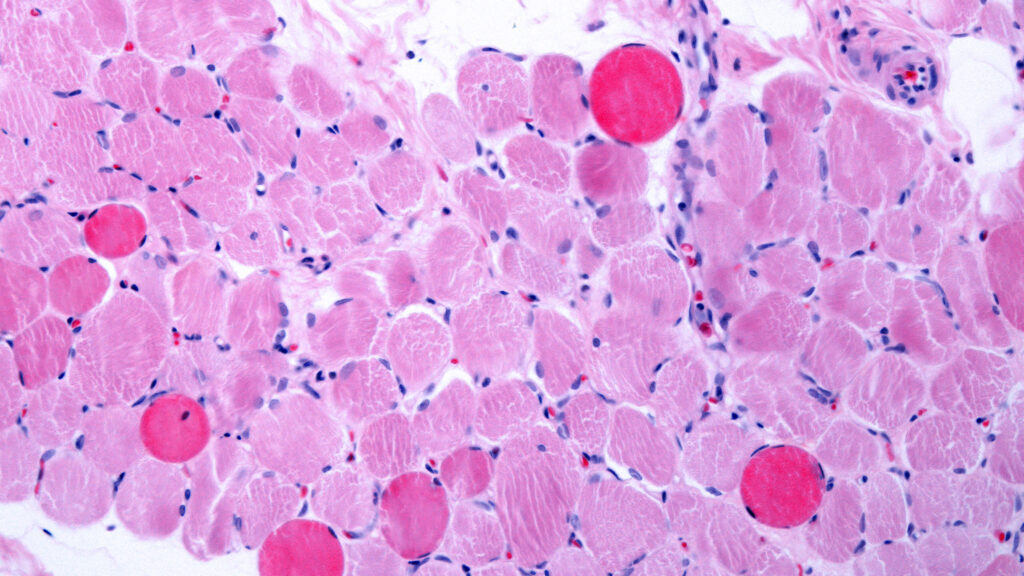
Capricor Therapeutics accused Nippon Shinyaku and its U.S. subsidiary of failing to follow through on marketing plans for a Duchenne muscular dystrophy treatment, and refusing to fix a pricing glitch that was belatedly discovered in their exclusive distribution agreement.
A key issue is a “fatal flaw” in a pricing formula that would make it “economically impracticable” for the therapy to reach patients covered by Medicare, Medicaid, and private insurers, according to a lawsuit filed in a New Jersey state court on Thursday. Nippon Shinyaku and NS Pharma, the subsidiary, disclosed the problem in March 2025.
Basically, the formula ties Medicare reimbursement to the price that Capricor would charge NS Pharma, since it would be the only U.S. buyer. But as it stands, the lawsuit indicated that health care providers would get reimbursed less than they would pay to cover the cost of acquiring and administering the medicine, which is called deramiocel.
Capricor Therapeutics accused Nippon Shinyaku and its U.S. subsidiary of failing to follow through on marketing plans for a Duchenne muscular dystrophy treatment, and refusing to fix a pricing glitch that was belatedly discovered in their exclusive distribution agreement.
A key issue is a “fatal flaw” in a pricing formula that would make it “economically impracticable” for the therapy to reach patients covered by Medicare, Medicaid, and private insurers, according to a lawsuit filed in a New Jersey state court on Thursday. Nippon Shinyaku and NS Pharma, the subsidiary, disclosed the problem in March 2025.
Basically, the formula ties Medicare reimbursement to the price that Capricor would charge NS Pharma, since it would be the only U.S. buyer. But as it stands, the lawsuit indicated that health care providers would get reimbursed less than they would pay to cover the cost of acquiring and administering the medicine, which is called deramiocel.
Uncategorized
Trump signs off on plan to oust embattled FDA Commissioner Marty Makary: report
If ultimately confirmed, Makary’s planned departure, broken by The Wall Street Journal Friday afternoon, would follow a controversial tenure in which his deputy and constant co-author Vinay Prasad riled biopharma feathers with myriad unexpected drug rejections. Prasad stepped down as biologics chief last week.
Uncategorized
FDA Commissioner Makary to be fired, WSJ reports
President Donald Trump is planning to fire FDA Commissioner Marty Makary, the Wall Street Journal reported on Friday.
The plans haven’t been finalized and could still change, the Journal said. Representatives …
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars