Uncategorized
Spatial Biology
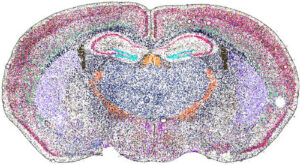
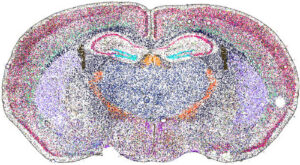
Spatial biology is a rapidly advancing discipline that examines biological molecules (such as DNA, RNA, and proteins) within their native locations in tissues. This approach offers critical insight into how cancer and immune cells interact within the tumor microenvironment.
Beginning in the 20th century, techniques such as immunohistochemistry and in situ hybridization allowed researchers to localize proteins and nucleic acid sequences directly in tissue samples. However, these analyses were typically limited to a single marker at a time. More recently, advances in high-throughput sequencing and mass spectrometry enable the simultaneous analysis of hundreds, or even thousands, of molecules while preserving spatial context. To explore the future of spatial biology, we spoke with four companies pioneering these technologies.
Spatial transcriptomics
The transcriptome is the complete collection of RNA produced in a cell or tissue at a given time. It reflects active gene expression, which varies with disease state and environmental conditions. Understanding the transcriptome is therefore crucial for studying cancer biology and treatment response.
Pratheesh Sathyan, PhD, head of AMR oncology at Illumina, says that the company plans to roll out a new spatial transcriptomics platform for commercial release in 2026. He describes several advantages of spatial transcriptomics.
“Immune cells are an important part of our defense against cancer. And spatial approaches help to understand how immune cells interact with the tumor,” he says. “With spatial transcriptomics, you are maintaining the true habitat of the tissue, and that helps us with better diagnostics and better precision therapy.”
Sathyan cites pancreatic cancer as an example. “In pancreatic cancer, spatial technology has shown these little regions of protected cancer cells that allow for the progression of the disease. Understanding these areas is especially important for difficult-to-treat tumors like pancreatic cancer, where the biology is very complex.”
The term “spatial transcriptomics” is about 10 years old, notes Darren Segale, PhD, senior director of scientific research at Illumina. He recalls how Nature Methods named spatially resolved transcriptomics as its Method of the Year for 2020.1
Since then, a great deal of progress has been made in the field. Segale notes that Illumina’s platform will enable sampling of more than seven square centimeters of tissue. “This allows you to look at large bits of tumor microenvironments. More than other products available today,” he says.
Another key feature of the upcoming product is whole transcriptome coverage. “Many spatial transcriptomics platforms look at panels of typically 5,000 unique transcripts,” notes Segale. “In contrast, we are seeing 30,000 to 40,000 unique RNA transcripts per sample. It is completely unbiased and hypothesis-free. There is a real richness of information that we are seeing.” Finally, the platform will enable 1-micron spatial resolution, allowing researchers to see key surface features on the cellular level.
As part of the workflow, the tissue will undergo standard hematoxylin and eosin (H&E) staining and imaging. The spatial assay can then be performed on the bench with any Illumina sequencer. Secondary analysis is accomplished with Illumina’s DRAGEN software.
software.
Sathyan highlights Illumina’s workshop at the 2025 Advances in Genome Biology and Technology (AGBT) conference, during which researchers from St. Jude’s Children’s Research Hospital shared data from the new platform. The research involved advanced prostate cancer.
“Using our spatial transcriptomics platform, they were able to find biomarkers that were not included in other targeted approaches,” says Sathyan. “This improved transcript coverage allowed for the analysis of rare cell types that were correlated with disease states.”
Illumina is applying its new platform to colorectal cancers, prostate cancer, and gliomas. The company presented additional breast cancer results at the 2026 AGBT conference.
“The information from our new platform will allow us to differentiate different cell types from one another and even identify new markers and new subtypes,” says Segale. “This is important because much of the cellular structure of the human body still has not been mapped, including organs and various tissues.”
They note that Illumina’s spatial transcriptomics platform is the company’s first in this product category. “We are launching this as part of a multi-omics approach in 2026 to understand cancer biology better. This will include single-cell, spatial, methylome, and proteomics analysis,” notes Sathyan.
Shifting toward the clinic
About one in every 20 people in the United States is living with Barrett’s esophagus, a condition caused by long-term exposure to stomach acid that changes the lining of the esophagus, notes Castle Biosciences’ vice president of research & development, Rebecca Critchley-Thorne, PhD. “While most people with this condition will never develop esophageal cancer, a small percentage of them do, even after appearing low risk on routine tissue examination,” she notes.
Traditional surveillance for Barrett’s esophagus relies on periodic endoscopies, followed by manual microscopic review of biopsies. “Unfortunately, this approach does not reliably distinguish patients who will remain stable from those who will progress to esophageal cancer,” says Critchley-Thorne.
Castle Biosciences’s TissueCypher spatial proteomics platform helps address this gap by identifying high-risk biological signals that may be present before cancer develops. “Measuring proteins within their spatial context in tissue makes it possible to detect subtle, complex patterns of disease biology that cannot be captured through traditional microscopy or bulk molecular tests alone,” notes Critchley-Thorne.

The TissueCypher test employs multiplex immunofluorescence, which utilizes antibodies labeled with fluorophores to simultaneously detect multiple markers. The patient’s tissue is labeled with fluorescent tags for nine protein biomarkers and then digitally imaged. These nine protein biomarkers are associated with key pathways in the neoplastic progression to esophageal adenocarcinoma—including tumor suppression, cell cycle control, angiogenesis, and immune cell infiltration.
The platform uses AI-driven recognition to identify relevant Barrett’s tissue and remove artifacts in tissue sections or slide mounting media. This is followed by image analysis to assess how proteins and cells are organized and interacting within the tissue.
Ultimately, the spatially resolved measurements are combined into a fixed algorithm that generates a risk score, placing patients into risk categories (high, intermediate, or low), and estimating the probability of disease progression to esophageal cancer over five years. Castle delivered more than 39,000 test reports in 2025.
Rather than replacing pathology, the TissueCypher test is designed to complement it by providing biological insight beyond what microscopic review can capture, stresses Critchley-Thorne.
Importantly, the early identification of at-risk patients creates the opportunity to intervene early with preventative treatments and more routine endoscopies. “By reducing the chance that high-risk patients progress undetected, this approach has the potential to prevent progression to esophageal cancer.”
Like Castle Biosciences, Discovery Life Sciences also offers multiplex immunofluorescence services, says Anna Juncker-Jensen, PhD, vice president of scientific affairs.
The company’s technology comprises two key instruments: the Akoya PhenoImager HT enables the rapid, high-throughput analysis of up to eight markers, while the Lunaphore COMET provides comprehensive, high-resolution profiling of up to 40 markers, including both RNA and protein.
HT enables the rapid, high-throughput analysis of up to eight markers, while the Lunaphore COMET provides comprehensive, high-resolution profiling of up to 40 markers, including both RNA and protein.
“Both platforms offer a variety of pre-characterized panels that enable the identification of distinct immune cell populations and a characterization of the immune landscape,” she says.
Juncker-Jensen highlights the company’s expertise in building validated, multiplexed panels customized for specific research projects. “These services are increasingly important tools for our biotech and pharma clients in the search for novel prognostic and predictive biomarkers for personalized cancer therapy. We also support regulatory-compliant workflows and quantitative endpoints for clinical trials.”
Discovery’s multiplexed panels enable analysis of spatial distribution patterns within the tumor microenvironment—such as the localization of lymphocytes, myeloid cells, fibroblasts, and vessels. They also elucidate key pathways in cancer progression, including changes in the migratory and invasive properties of cells, says Juncker-Jensen.
Ultimately, Critchley-Thorne envisions that multiplexed immunofluorescence platforms will increasingly be applied in medicine. “This detailed tissue information moves care toward more personalized, biology-driven decisions that can ultimately benefit patients across a range of diseases.”
Bringing in new omics
Mike Easterling, PhD, vice president of MALDI Imaging at Bruker Daltonics, stresses that he is interested in more than just RNA and proteins.
“We often hear multi-omics defined as transcriptomics and proteomics. That leaves critical biochemical layers unexplored, and this is where mass spectrometry contributes. We integrate metabolomics, glycomics, and lipidomics because these molecular classes are responsible for disease mechanisms that transcriptomics alone cannot capture.”
“Say you need to co-localize five transcripts, seven proteins, and several glycans and lipids within the same tissue region, organized by their spatial relationship to one another. That multi-omic biomarker composite may define a disease state much more precisely than any single layer. Our platforms support exactly that kind of investigation.”

Bruker uses matrix-assisted laser desorption/ionization (MALDI) mass spectrometry as the foundation of its multi-omics approach.
“Mass spectrometry is inherently multiplexed in a way that antibody-based approaches aren’t. A single acquisition can simultaneously detect hundreds of lipid species, glycans, and metabolites across a tissue section without the need for target-specific labels. That is a fundamental advantage for unbiased discovery.”
Easterling emphasizes that Bruker’s approach is an ideal way to capture information about post-translational modifications (PTMs). Such information can be challenging to obtain reliably with antibody-based workflows, he adds.
PTMs, particularly glycosylation, are directly implicated in a host of processes like immune evasion, tumor progression, and metastasis, he notes.
“Given what we know, glycans are among the most structurally complex PTMs, exhibiting enormous diversity in composition. Unlike proteins, glycan structures are not directly encoded by the genome, making them difficult to probe with targeted assays. Mass spectrometry offers a way to profile this complexity directly from tissue.”
Easterling recounts a collaboration with Stanford University and the Medical University of South Carolina to spatially localize glycans in different tissues. This information was then combined with spatial transcriptomics and proteomics layers to analyze gliomas, a lethal cancer with very limited treatment options. Ultimately, the glycomics information emerged as the strongest classifier of tumor grade across all three omics layers.
Bruker is also collaborating with the Medical University of South Carolina to investigate how PTMs are distributed throughout the extracellular matrix using MALDI imaging. “Research usually focuses on the cell as the fundamental unit of disease biology, but the extracellular matrix is increasingly recognized as a critical regulator of a number of processes related to tumor progression,” he explains.
Spatial proteomics and transcriptomics are often quite expensive. In contrast, Easterling stresses the cost-effectiveness of Bruker’s MALDI-based techniques. With MALDI imaging, he notes that the resolution typically ranges from five to 20 microns. Running at a higher resolution (five microns) typically takes longer and increases costs and laboratory time.
“In discovery, your goal is comprehensive molecular characterization. Running at five-micron resolution provides the spatial detail needed to resolve individual cell populations. With smaller cohort sizes, longer acquisition times are acceptable.”
“But on the translational side, you typically know what you’re looking for. Operating at 10 to 20 microns significantly reduces acquisition time and cost per sample while still providing the spatial acuity required for clinically relevant readouts,” he says.
Reference
1. Method of the year 2020: spatially resolved transcriptomics. Nat Methods. 2021;18(1):1. doi:10.1038/s41592-020-01042-x.
The post Spatial Biology appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
BioNTech to shutter Singapore HQ after ‘comprehensive review’
BioNTech, in a move to streamline its operations, is set to close its factory in Singapore that it bought from Novartis just over three years ago.
The facility at the Tuas Biomedical Park, which employs …
Uncategorized
STAT+: Merck’s experimental HIV prevention pill could be made for less than $5 a year, researchers say
An experimental HIV prevention pill being developed by Merck could be mass produced for less than $5 per patient a year according to a new analysis. Advocates argue the low cost means the company should find it easier to license the drug so that low- and middle-income countries can gain easy access.
The pill, dubbed MK 8527, is currently undergoing a pair of late-stage clinical trials that are expected to determine whether the medicine can lower HIV transmission when given to people at high risk of infection. The results are due in the latter half of 2027, according to ClinicalTrials.gov.
Already, the pill is generating considerable interest after Merck released mid-stage results last summer showing its drug holds promise. In addition to being safe and effective, the study found it could protect against infection, a form of prevention known as pre-exposure prophylaxis or PrEP, within 24 hours after being taken. Merck noted the pill works in a novel way.

An experimental HIV prevention pill being developed by Merck could be mass produced for less than $5 per patient a year according to a new analysis. Advocates argue the low cost means the company should find it easier to license the drug so that low- and middle-income countries can gain easy access.
The pill, dubbed MK 8527, is currently undergoing a pair of late-stage clinical trials that are expected to determine whether the medicine can lower HIV transmission when given to people at high risk of infection. The results are due in the latter half of 2027, according to ClinicalTrials.gov.
Already, the pill is generating considerable interest after Merck released mid-stage results last summer showing its drug holds promise. In addition to being safe and effective, the study found it could protect against infection, a form of prevention known as pre-exposure prophylaxis or PrEP, within 24 hours after being taken. Merck noted the pill works in a novel way.
Uncategorized
Opinion: STAT+: Former Geisinger CEO: U.S. health systems must replace huge numbers of people with AI
About 20 years ago, I stepped on stage at one of our Geisinger town halls and looked out upon a sea of people: thousands of full-time employees at an integrated health system charged with the health and well-being of millions of Pennsylvanians.
Only a fraction of the people in that room were clinicians.
That was the first time I fully visualized the problem: We employed more people in our revenue cycle department to process bills and reconcile data than we did doctors. And we weren’t alone. It’s the same story at every health system in America, large and small, and over the past two decades, the ratio has become dramatically more disparate.

About 20 years ago, I stepped on stage at one of our Geisinger town halls and looked out upon a sea of people: thousands of full-time employees at an integrated health system charged with the health and well-being of millions of Pennsylvanians.
Only a fraction of the people in that room were clinicians.
That was the first time I fully visualized the problem: We employed more people in our revenue cycle department to process bills and reconcile data than we did doctors. And we weren’t alone. It’s the same story at every health system in America, large and small, and over the past two decades, the ratio has become dramatically more disparate.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors