Uncategorized
New Single‑Cell Platform Tracks RNA and Protein in Immune Signaling
A new single‑cell sequencing method is giving researchers a clearer view of how immune cells actually behave—capturing not just what they plan to do, but what they are doing in real time. The platform, called CIPHER‑seq, measures RNA and proteins simultaneously inside the same immune cell, exposing gaps between genetic intent and functional output that have long complicated studies of cytokine signaling. The work, titled “CIPHER-seq enables intracellular multimodal profiling of cytokine responses in single immune cells,” appears in Scientific Reports.
Single‑cell RNA sequencing has reshaped immunology by revealing which genes are switched on across thousands of cells at once. But RNA alone can be misleading, especially for cytokines. However, RNA is only a set of instructions; proteins carry out the action. And for cytokines, RNA levels often fail to predict how much protein a cell actually produces. “In immune cells, RNA and protein don’t always rise and fall together,” said co‑senior author Emiliano Cocco, PhD, an assistant professor of biochemistry and molecular biology at the Miller School.
CIPHER‑seq (Cytokine Intracellular Protein High-throughput Expression with RNA-sequencing) was designed to close that gap. Developed by researchers at the Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine, together with collaborators at UCSF and the Helen Diller Family Comprehensive Cancer Center, the method gently preserves cells and captures multiple molecular layers at once. From a single immune cell, CIPHER‑seq can quantify genome‑wide RNA, surface proteins, intracellular proteins, and cytokines that have not yet been released—creating a more complete snapshot of immune activity than RNA‑only approaches.
“RNA gives us clues about where a cell is headed,” said co‑senior author Justin Taylor, MD, a Sylvester physician-scientist. “Proteins show us where it actually arrives, and this clearer picture could help scientists design better immunotherapies and help clinicians predict which patients are most likely to benefit from them.”
The team validated the platform by stimulating peripheral blood mononuclear cells (PMBCs) and tracking their responses. According to the study, CIPHER‑seq captured robust induction of key cytokines—including interferon‑gamma and tumor necrosis factor—while also resolving metabolic remodeling during activation. Importantly, the method revealed the timing of these events: RNA signals rose first, followed by delayed but consistent protein accumulation. First author Avni Bhalgat, PhD, described it as “seeing the plan before the action. Cytokines help determine whether immune cells attack cancer, ignore it, or even help tumors grow.”
The researchers also compared CIPHER‑seq with standard single‑cell workflows and found a notable difference: cells processed with CIPHER‑seq showed far fewer mitochondrial stress signatures. Some existing protocols inadvertently damage cells during preparation, triggering artificial stress responses. By reducing these artifacts, CIPHER‑seq provides a cleaner readout of immune behavior.
The authors emphasize that this multimodal view is especially valuable for studying cancer, inflammation, and treatment resistance—contexts where cytokine timing and protein abundance can shape therapeutic outcomes. “The platform helps us move beyond inference and toward understanding how immune responses truly unfold—one cell at a time,” Taylor added. By tracking RNA and protein together, CIPHER‑seq moves researchers beyond inference and toward a step‑by‑step understanding of how immune responses unfold.
The post New Single‑Cell Platform Tracks RNA and Protein in Immune Signaling appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
STAT+: Key GOP senators push back on Trump’s plan to cut NIH, reorganize HHS

WASHINGTON — Both Democratic and Republican Senators who oversee federal spending seemed skeptical of proposed cuts to health research and public health in the White House’s budget, potentially teeing up a congressional package that ignores many of the administration’s most dramatic proposals for a second year.
During a Senate appropriations health subcommittee hearing Tuesday, lawmakers questioned health secretary Robert F. Kennedy Jr. on how his department could tackle chronic disease, smoking cessation, and cancer research with a proposed 2027 fiscal budget that would cut the department by 12%.
The request, which is broadly similar to what was proposed last year, includes deep cuts to the National Institutes of Health, the elimination of a health research agency, and the creation of a new agency devoted to chronic diseases called the Administration for a Healthy America.
Uncategorized
Medicare indefinitely delays pilot plan to cover weight loss drugs
Medicare indefinitely delays pilot plan to cover weight loss drugs
The Centers for Medicare and Medicaid Services has delayed a key pilot program focused on Medicare coverage for obesity medications, after insurance plans indicated they wouldn’t participate at this time.
Abe Sutton, head of the … Read More
Uncategorized
Alzheimer’s Linked to Cancer Mutations in Brain Immune Cells
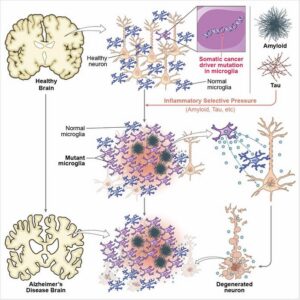
As the body ages, cells naturally accumulate dozens of genetic mutations each year. New research reported by researchers at Boston Children’s Hospital suggests that the brain’s resident immune cells, microglia, amass mutations in specific cancer-driving genes, yet they don’t manifest as cancer. Instead, these mutations may help drive Alzheimer’s disease.
The research team, led by Christopher Walsh, MD, PhD, chief of the Division of Genetics and Genomics at Boston Children’s and an investigator of the Howard Hughes Medical Institute, and collaborators Alice Eunjung Lee, PhD, and August Yue Huang, PhD, also in the Division of Genetics and Genomics—who are all professors at Harvard Medical School and associate members of the Broad Institute of MIT and Harvard—say their study findings may provide insights into new Alzheimer’s disease diagnostics and treatments.
“We find that to some extent, Alzheimer’s disease is a little like cancer—driven by the same mutations that drive blood cancers like lymphoma and leukemia,” said Walsh. “This is helpful because we have a lot of drugs to fight cancer and some of them might be useful therapeutically for Alzheimer’s disease.”
The researchers reported on their work in Cell, in a paper titled “Somatic cancer variants enriched in Alzheimer’s disease microglia-like cells drive inflammatory and proliferative states.”
Microglia function as the brain’s resident immune cells, acting as garbage collectors, eating debris and infected or dying cells. “The importance of microglia in Alzheimer’s disease (AD) pathogenesis has been demonstrated by large-scale genetic association studies, which have identified AD risk variants in a growing list of microglia-related genes,” the authors wrote. “Once abnormally reactive in AD, microglia can promote synaptic and neuronal loss while exacerbating tau proteinopathy.”
Unlike the rest of the immune system cells that circulate in the blood throughout the body, microglia don’t cross the blood brain barrier—or so experts thought. For their newly reported study the research team sequenced 149 cancer-driving genes from tissue samples in 190 brains donated from people with Alzheimer’s disease compared to 121 healthy brains. The Alzheimer’s samples had more single DNA letter changes than the healthy tissue with the most changes found repeatedly in the same five cancer driver genes, meaning the microglia were amassing mutations in specific genes. “Deep (>1,000×) panel sequencing of 311 brain samples revealed enrichment of somatic single-nucleotide variants (sSNVs) in cancer driver genes in AD brains, especially in genes associated with clonal hematopoiesis (CH),” the team stated.
The cancer gene mutations the researchers discovered in the microglia are commonly found in blood cancers. Because of this, the team tested blood samples from people with Alzheimer’s disease for these same mutations. The team didn’t expect the blood to have these mutations. However, Walsh’s team found the blood cells of the same Alzheimer’s patients carried the same cancer mutations too.
![Microglia-like immune cells with cancer mutations (purple) emerge in the brain. Separately, clumps of proteins, like Tau or amyloid, accumulate in the brain, making the environment hostile. Those microglia cells with mutations get selected for survival and proliferation, creating an inflammatory environment that makes innocent bystander neurons die, contributing to Alzheimer’s disease. [Christopher Walsh and colleagues at Boston Children's Hospital]](https://www.genengnews.com/wp-content/uploads/2026/04/Low-Res_Walsh-Cell-photo-300x300.jpg)
“These sSNVs were associated with clonal expansion and carried by both microglia-like brain macrophages (MLBMs) in multiple brain regions as well as paired blood, suggesting a likely hematopoietic origin,” the investigators stated. “It was actually a really unexpected finding that suggests a totally new mechanism for Alzheimer’s disease pathogenesis,” said Huang. “The findings mean that the blood’s immune cells with cancer mutations are likely getting into the brain and contributing to disease.”
The researchers theorize that the blood-brain barrier weakens, either by age or injury, allowing the blood’s immune cells to cross into the brain. These new arrivals then convert into microglia-like cells. Separately, clumps of proteins accumulate in the brain, triggering microglia to proliferate and respond. The cells most likely to dominate are those with a selective advantage, such as the microglia-like cells with the cancer mutations. However, these mutant microglia also make the environment more inflammatory and hostile than that of the healthy microglia, causing innocent bystander neurons to die off, which leads to Alzheimer’s disease. “These findings suggest that clonal somatic driver variants in MLBMs are enriched in AD, potentially promoting neuroinflammation and neurodegeneration,” the researchers noted. “Potential roles of somatic cancer driver variants in AD pathogenesis open up a whole new range of therapeutic avenues in AD, complementary to approaches emphasizing amyloid and tau.”
Lee added, “Because it’s hard to access brain tissue in a living patient, genetic screens using blood samples could be developed to test whether a person carries these mutations, and has an increased risk of developing Alzheimer’s disease.” Lee and Huang performed a follow-up study, now posted as a preprint on bioRxiv. Here, they demonstrated that cancer driver mutations observed in patient blood samples increased risk of Alzheimer’s disease independently of a well-established genetic risk factor, APOE4.
The post Alzheimer’s Linked to Cancer Mutations in Brain Immune Cells appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars




