Uncategorized
Lilly to Acquire Kelonia for Up to $7B, Expanding Cancer Cell Therapy Pipeline
Eli Lilly has agreed to acquire Kelonia Therapeutics for up to $7 billion, the companies said today, in a deal that would bolster the buyer’s oncology pipeline with an early clinical phase lentiviral in vivo chimeric antigen receptor T-cell (CAR T) therapy under study in relapsed/refractory multiple myeloma.
Kelonia’s lead program KLN-1010 is a one-time intravenous gene therapy designed to generate anti-B-cell maturation antigen (BCMA) CAR T cells, targeting the BCMA protein expressed on the surface of multiple myeloma cells.
In December at the American Society of Hematology (ASH) 2025 Annual Meeting, Kelonia presented positive early clinical data for KLN-1010 from the Phase I inMMyCARTM trial (NCT07075185). The data showed the CAR T therapy to have 100% minimal residual disease (MRD)-negative response rate across four patients, all of whom remained in response through the longest follow up of five months.
Those and other results, according to the company, provided initial clinical validation of KLN-1010 and demonstrated promising tolerability. In January, Kelonia won FDA clearance for an investigational new drug (IND) application for KLN-1010, enabling the trial to expand from Australia into multiple clinical sites across the United States.
“The early clinical data for KLN-1010 are highly encouraging, both as a potential step forward for patients with multiple myeloma and as proof of concept for Kelonia’s platform,” Jacob Van Naarden, executive vice president and president of Lilly Oncology and head of corporate business development, said in a statement.
Investors appeared more sanguine about the Kelonia acquisition as Lilly shares were all but flat in early Monday trading as of 11 a.m. ET, to $927.16 from Friday’s close of $927.03. Kelonia is privately held.
KLN-1010 applies the company’s in vivo gene placement system (iGPS®), which uses engineered lentiviral-based particles designed to efficiently and selectively enter T-cells inside the body, enabling a patient’s own body to generate CAR T therapies designed to treat underlying disease.
Lilly and Kelonia reason that KLN-1010 could transform treatment of multiple myeloma by eliminating challenges associated with both ex vivo patient-specific cell therapy manufacturing, and pre-administration chemotherapy.
“Autologous CAR T therapies have meaningfully improved outcomes for patients with various cancers, but significant manufacturing, safety, and access barriers mean that only a fraction of eligible patients actually receive them,” Van Naarden added. “Kelonia’s in vivo platform has the potential to change that by delivering rapid, durable responses in a far simpler, off-the-shelf format.”
Kelonia marks Eli Lilly’s fourth announced acquisition of a smaller biotech this year:
- In March, Lilly committed up to $7.8 billion to acquire Centessa Therapeutics, a developer of sleep disorder drugs.
- A month earlier, Lilly announced plans to buy out circular RNA cell therapy developer Orna Therapeutics for up to $2.4 billion, targeting advancements in cell therapy.
- And in January, Lilly inked a $1.2 billion acquisition of Ventyx Biosciences, an NLRP3-targeting oral drug developer focused on inflammatory diseases.
Behind the deals
Behind all the deals is the pharma giant’s desire to capitalize on the billions of dollars it is generating from sales of its obesity and diabetes drugs based on glucagon-like peptide 1 (GLP-1) receptor analysts alone or in tandem with a glucose-dependent insulinotropic polypeptide (GIP). Lilly markets tirzepatide, a GLP-1/GIP dual agonist, in obesity as Zepbound® ($13.542 billion in 2025 sales) and in diabetes as Mounjaro® ($22.965 billion).
Lilly stands to generate even more in obesity-related sales in coming years once it brings to market its oral obesity drug Foundayo (orforglipron), a small molecule GLP-1 receptor agonist—though analysts predict the drug’s 2026 sales will likely be lower than once expected because of the price war Foundayo faces competing head to head with Lilly’s arch-rival in obesity drugs, Novo Nordisk. In December, Novo Nordisk got a jump on Lilly when the Danish biotech giant won FDA approval for oral Wegovy® (semaglutide), a once-daily 25 mg GLP-1 receptor agonist tablet indicated for chronic weight management.
(orforglipron), a small molecule GLP-1 receptor agonist—though analysts predict the drug’s 2026 sales will likely be lower than once expected because of the price war Foundayo faces competing head to head with Lilly’s arch-rival in obesity drugs, Novo Nordisk. In December, Novo Nordisk got a jump on Lilly when the Danish biotech giant won FDA approval for oral Wegovy® (semaglutide), a once-daily 25 mg GLP-1 receptor agonist tablet indicated for chronic weight management.
A Lilly buyout of Kelonia could compel Johnson & Johnson to take a closer look at acquiring Legend Biotech, Kostas Biliouris, PhD, a managing director on the biotechnology research team of Oppenheimer, wrote Sunday in a research note. He cited the fact J&J’s Janssen Biotech successfully partnered with Legend to develop Carvykti® (ciltacabtagene autoleucel), a B-cell maturation antigen (BCMA)-directed CAR T-cell therapy indicated for adults with relapsed or refractory multiple myeloma who have received at least one prior line of therapy. Carvykti generated $1.877 billion in sales last year, up nearly double (96%) from $963 million in 2024.
Also, Biliouris cited the presence in Legend’s pipeline of LUCAR-G39D, a clinical in vivo CAR T program designed to treat B-cell non-Hodgkin’s lymphoma by targeting CD19xCD20. LUCAR-G39D showed positive first-in-human safety and efficacy data from a Phase I trial (NCT06395870) at ASH last December.
“We believe in vivo CAR T technology has strong potential, as treatment process is fast and circumvents the need for lymphodepletion, but think it will likely take ~6-8years before safety/durability questions are addressed, and regulatory approval is granted,” Biliouris predicted.
Lilly has agreed to acquire Kelonia for $3.25 billion upfront plus up to $3.75 billion in future payments tied to achieving specified clinical, regulatory, and commercial milestones. The acquisition deal is subject to regulatory approvals and other customary closing conditions, and is expected to close in the second half of 2026.
Upon closing, Lilly said, it will determine how to account for the transaction in accordance with Generally Accepted Accounting Principles (GAAP), then reflect the deal in future financial results and financial guidance.
“Kelonia’s leadership in advancing the immense promise of in vivo cell therapy is unmatched, extending its reach and impact beyond the traditional boundaries of personalized medicine,” Kelonia CEO Kevin Friedman, PhD, stated. “We have demonstrated the ability to achieve deep multiple myeloma remissions with significantly reduced complexity and cost relative to ex vivo CAR T-cell approaches.”
“In combination with Lilly’s strengths, our in vivo iGPS platform is positioned to broaden the reach of cell therapy beyond the current CAR T landscape in hematologic malignancies and to transform treatment across a far wider range of cancers and other serious diseases,” Friedman added.
The post Lilly to Acquire Kelonia for Up to $7B, Expanding Cancer Cell Therapy Pipeline appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Synthetic Biology and Tissue Engineering Grow Liver Tissue In‑Body
Damage to the liver in patients developing end-stage liver disease has become too severe for the organ’s normally extraordinary regenerative capacity to repair or compensate for that damage. Once this point of no return has been reached the only option is an organ transplant. However, donor livers are in high demand and very limited supply.
Ambitious efforts are on the way that eventually could enable the engineering of entire implantable liver organs. However, the maximum size of laboratory-engineered liver constructs remains limited and cannot yet provide therapeutic benefits for patients. A research team at the Wyss Institute at Harvard University, Boston University, and MIT has now approached this important problem from a different angle.
“We asked if it would be possible to first implant a small-scale liver construct and then drive it to expand in the body following its engraftment,” said Christopher Chen, MD, PhD, a Wyss Institute core faculty member and the William Fairfield Warren Distinguished professor of biomedical engineering and director of the Biological Design Center at Boston University. “A sufficiently grown, functional ‘satellite liver’ could immediately relieve the metabolic burden in a damaged liver and help bridge the time until a transplant becomes available.”
Chen co-led the research together with associate faculty member Sangeeta Bhatia, MD, PhD, who is the John J. and Dorothy Wilson Professor of Health Sciences and Technology and of Electrical Engineering and Computer Science at the Koch Institute for Integrative Cancer Research at MIT, and a Howard Hughes Medical Institute investigator. Chen is also a leader of the Wyss Institute’s 3D Organ Engineering Initiative, and team lead of the recently awarded ARPA-H PRINT-supported ImPLANT project, which focuses on whole organ liver engineering at the Wyss and collaborating institutions.
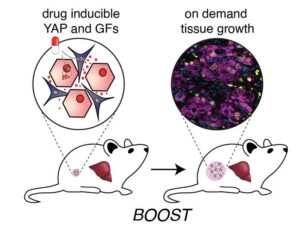
The project, spearheaded by Amy Stoddard, PhD, (MIT ’25), who developed the approach in her doctoral research and then as a postdoctoral fellow, integrates tissue engineering and synthetic biology tools in a genetic strategy the team has named “bioengineered on-demand outgrowth via synthetic biology triggering,” or BOOST. By specifically rewiring the gene expression of primary liver hepatocytes and supportive fibroblast cells, the scientists were able to effectively switch on a tissue growth program in small, engineered liver constructs after their implantation into mice.
“Using engineered liver tissue as a proof-of-concept application, we integrated synthetic biology and tissue engineering tools to build liver tissues that can be expanded on-demand after implantation in vivo,” the team reported in their published paper in Science Advances, which is titled “Synthetic control of implanted engineered liver tissue growth.” In the paper they concluded “In this study, we define the first steps toward an unconventional approach to cell therapy scale-up: engineering a small construct and then inducing it to grow in situ … “This strategy, which we have named BOOST, could provide several key advantages, including circumventing the need for large quantities of cellular raw materials and bypassing the formidable challenge of generating a rapidly perfusable construct that can survive the engraftment period.”
The authors wrote, “Organ transplant is currently the only curative treatment for patients with end-stage organ failure, yet this therapy is inaccessible to many due to the paucity of organs available for transplant.” And while significant progress has been made in the field of engineering tissue-based cell therapies that could represent alternatives, or bridges to transplant, they acknowledge, “… scaling of these constructs to sizes of therapeutic relevance remains a barrier to clinical translation.”
In order to address current challenges associated with fabrication, Chen and colleagues looked at the problem from different angle, asking whether it would be possible to first implant a small-scale construct and then trigger it to expand in situ, after its engraftment into the host.
To be able to induce growth of an implanted small liver constructs in situ within a recipient’s body the researchers first needed to identify the relevant cues that would allow them to do so. “A key first step toward this method of in situ scale-up would be the successful control of cellular growth within the engineered construct after engraftment,” they wrote. Since liver growth is known to be regulated by soluble growth factors (GFs), Stoddard screened a collection of candidate factors to identify those that, when added to cultured human primary hepatocyte cells (HEPs), had the strongest growth-inducing effects.
![The genetic “BOOST” strategy integrates tissue engineering and synthetic biology tools to enable on-demand liver growth inside the body. By specifically rewiring the gene expression of primary liver hepatocytes and supportive fibroblast cells, a tissue growth program is switched on in a small, engineered liver construct after its implantation into recipients and upon addition of an inducing agent (shown as a pill). As a result, the hepatocytes in the construct start and continue to proliferate until a desired construct size has been reached and the inducing signal is not provided anymore. In mice, BOOST resulted in robust and healthy liver growth. [Wyss Institute at Harvard University]](https://www.genengnews.com/wp-content/uploads/2026/04/Low-Res_Press-graphics-01-300x225.jpg)
“We ended up with a set of four growth factors, HGF, TGFa, WNT2 and RSPO3, that potently induced sparsely scattered HEPs to grow in the culture dish,” said Stoddard. “But when we tested whether they could do the same in 3D liver tissues consisting of densely packed HEPs and fibroblasts, they turned out to be ineffective. This led us to hypothesize that there must be an additional mechanism at work in human HEPs that inhibits cell proliferation in high-density conditions.”
The team homed in on a protein, YAP, that senses mechanical signals, and which was known to move from cells’ cytosol to their nucleus in low-density conditions to help express genes involved in cell proliferation. However, in high-density conditions when cells are compressed, YAP is degraded in the cytosol, which prevents the activation of those target genes and restricts proliferation.
“Importantly, when we overexpressed a non-degradable version of YAP in HEPs, which also reaches the nucleus in high-density conditions to partake in gene regulation, we successfully overrode this density checkpoint in HEPs,” Stoddard said. “Interestingly, we found that HEPs needed to be stimulated with both YAP and GFs in order to grow in densely packed 3D liver tissues.”
Toward the goal of safely inducing and controlling HEP proliferation in a living organism, and eventually human patients, the researchers deployed synthetic biology tools to locally install control of these signaling pathways in HEPs and fibroblast cells within the engineered 3D liver tissues themselves. “We set out to engineer a synthetic biology toolkit capable of locally modulating growth factor and YAP signaling within engineered liver tissue, enabling on-demand control of proliferation even after implantation,” they noted.
The team engineered fibroblast cell lines that each secreted one of the four GFs, and HEPs that expressed the non-degradable YAP protein. And they made the expression of all proteins doxycycline (DOX)-inducible. They determined in time course experiments that a continuous seven-day treatment with DOX led 3D liver tissue composed of engineered cells to robustly expand in size and cell numbers in the culture dish. On DOX removal the HEPs reverted back to a non-proliferating state.
However, Stoddard noted, “… when we compared the gene expression of single cells in BOOST-engineered, DOX-induced 3D liver tissue to that of cells in non-engineered or BOOST-engineered, non-induced 3D liver tissue, we noticed that the expansion came with a trade-off: high proliferation rates went hand in hand with a less functional HEP state. While we believe this is a natural trade-off seen in a wide variety of biological settings, we hope to be able to address this in the future, recognizing that the liver also has native re-functionalization signals to harness.”
The litmus test for BOOST-engineered growth in 3D liver tissues was to see whether they would similarly expand following their implantation into living mice that were systemically treated with DOX for the same seven-day duration. Experiments showed that the implanted tissue exhibited a striking 500% increase in proliferation with a doubling of the engineered HEPs alone, and was vascularized to accommodate the metabolic demands of the expanded tissue. The tissue implants were also well tolerated by the mice, with no signs of fibrosis due to invading immune cells and fibroblast inflammation, or of tumor growth.
“These results were particularly exciting to us,” said Stoddard. “Prior to our work, injury to the host liver has always been required to trigger hepatocyte engraftment and proliferation. Here we were able to relieve this dependence, and trigger on-demand growth of implanted liver tissue in a completely healthy host.”
In the future, the team will explore the capacity of BOOSTed liver tissue to rescue the host in the setting of liver injury. “Our BOOST strategy lays the foundation for a future when solid organ cell therapies can be controlled non-surgically according to the needs of patients and their physicians,” Bhatia noted. “Beyond treating liver disease, the premise of BOOST could be applied to other engineered tissue therapeutics that are similarly constrained by challenges associated with tissue scale-up, such as engineered heart or pancreatic tissue to address serious diseases.”
In their paper the authors concluded, “… this work serves as an exciting proof-of-concept demonstration that scale-up of tissues via growth could be possible … Together, this work helps lay the foundations for a future of ‘smart’ tissue therapeutics that can be scaled to a patient’s needs and thereby offer treatment for numerous, previously incurable, diseases.”
The post Synthetic Biology and Tissue Engineering Grow Liver Tissue In‑Body appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
STAT+: In early trial, CAR-T results raise hope of preventing multiple myeloma in high-risk patients
SAN DIEGO — Alison Cameron spent close to a decade fighting to keep myeloma under control. She’d been diagnosed with smoldering multiple myeloma, a precursor to cancer, and spent years receiving infusions to keep it from progressing to active multiple myeloma. Now, after receiving CAR-T therapy, an aggressive immunotherapy, while on a trial, the 54-year old anesthesiologist is hoping the risk of cancer is gone for good.
It’s a reasonable hope, given the results of that trial, which researchers presented at the American Association for Cancer Research meeting here on Monday. All 20 patients who received the trial treatment no longer had any detectable myeloma cells in their body. That’s a far deeper and more complete response than scientists typically expect when it comes to multiple myeloma, and it’s prompting some experts to consider the possibility these patients have truly had active cancer permanently averted.
Currently, there is only one approved therapy for high risk smoldering myeloma, an antibody therapy called Darzalex. Patients can remain on treatment for years, but without achieving these kinds of deep molecular responses, and many still progress within 5 years, said Ecaterina Dumbrava, a cancer researcher at MD Anderson Cancer Center who did not work on the study. “The results raise a very important question whether early immune interception can not only delay progression but redefine treatment goals. Can we talk about the word we always avoid, which is cure,” she said.
Uncategorized
Trump, FDA plan to expedite psychedelic development
Over the weekend, President Donald Trump endorsed the potential of psychedelic treatments and vowed to expedite their research and development as remedies for severe mental health conditions, a seminal moment for a field long shunned …
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars