Uncategorized
Landmark Pancreatic Cancer Trial Highlights Promise of RAS-Targeting Daraxonrasib
Earlier this week, Revolution Medicines reported positive results from a global Phase III trial of its RAS‑targeting inhibitor daraxonrasib (RMC-6236) in metastatic pancreatic ductal adenocarcinoma (PDAC). In the RASolute 302 trial, patients receiving daraxonrasib achieved longer progression‑free survival (PFS) and overall survival (OS) than those on standard cytotoxic chemotherapy.
The RASolute 302 trial enrolled patients with pancreatic tumors harboring a wide range of RAS variants, including those with RAS G12 mutations (such as G12D, G12V, and G12R), as well as those without an identified RAS mutation. The primary endpoints of the trial were PFS and OS in patients with tumors harboring RAS G12 mutations. Secondary endpoints assessed PFS and OS in all enrolled patients (the intent-to-treat population), including those with tumors with and without (wild type) an identified RAS mutation.
Daraxonrasib patients achieved a median OS of 13.2 months versus 6.7 months for chemotherapy. The drug was generally well tolerated, with a manageable safety profile and with no new safety signals.
“With these unprecedented results, daraxonrasib has the potential to achieve our goal of bending the mortality curve in pancreatic cancer. Unlike chemotherapy, daraxonrasib is a RAS-targeted medicine that targets RAS in its active ‘ON’ state, shutting down a key signaling pathway that drives aggressive tumor growth. This is especially important in pancreatic cancer, which is among the most RAS-driven cancers, with more than 90% of tumors harboring a RAS mutation that is the driver of the cancer,” asserted Mark A. Goldsmith, MD, PhD, CEO and chairman of Revolution Medicines.
Pancreatic cancer carries one of the highest mortality rates of any solid tumor, a consequence of late-stage diagnosis and resistance to standard chemotherapy. In the United States, recent estimates point to roughly 60,000 new cases and nearly 50,000 deaths each year. With most PDAC tumors driven by RAS alterations, the early success of emerging RAS‑targeted strategies hints at how much more may be possible as this therapeutic space continues to expand.
RAS is the key oncogenic driver of pancreatic cancer. Nearly all RAS mutations occur at KRAS position G12, but RAS mutations in other isoforms and at KRAS positions G13 and Q61 are also observed. Daraxonrasib works by suppressing RAS signaling through inhibition of the interaction between both wild-type and mutant RAS(ON) proteins and their downstream effectors.
Pancreatic cancer is the most RAS-addicted of all major cancers, with more than 90% of patients harboring tumors driven by mutations in RAS proteins. These mutations span a range of RAS variants that fuel aggressive tumor behavior. Daraxonrasib, a multi-selective inhibitor of RAS(ON) proteins, is the first investigational agent in a novel class of RAS inhibitors designed to address a diverse and broad spectrum of oncogenic RAS drivers.
“For patients with metastatic pancreatic cancer, new treatment options are urgently needed to increase survival time and improve quality of life,” said Brian M. Wolpin, MD, MPH, professor of medicine at Harvard Medical School, director of the Hale Family Center for Pancreatic Cancer Research at Dana-Farber Cancer Institute, and principal investigator for the RASolute 302 trial. “The widely anticipated results of this study indicate that daraxonrasib provides a clear and highly meaningful step forward for patients with pancreatic cancer who have experienced progression on prior treatment, typically chemotherapy. I believe that this new approach is a very important advance for the field that I expect will be practice-changing for physicians and improve the care for patients with previously treated metastatic pancreatic cancer.”
Revolution Medicines now intends to submit the drug for approval by regulatory authorities, including the U.S. Food and Drug Administration as part of a future New Drug Application, and for presentation at the 2026 American Society of Clinical Oncology Annual Meeting.
The post Landmark Pancreatic Cancer Trial Highlights Promise of RAS-Targeting Daraxonrasib appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Alzheimer’s Linked to Cancer Mutations in Brain Immune Cells
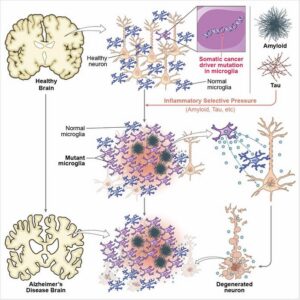
As the body ages, cells naturally accumulate dozens of genetic mutations each year. New research reported by researchers at Boston Children’s Hospital suggests that the brain’s resident immune cells, microglia, amass mutations in specific cancer-driving genes, yet they don’t manifest as cancer. Instead, these mutations may help drive Alzheimer’s disease.
The research team, led by Christopher Walsh, MD, PhD, chief of the Division of Genetics and Genomics at Boston Children’s and an investigator of the Howard Hughes Medical Institute, and collaborators Alice Eunjung Lee, PhD, and August Yue Huang, PhD, also in the Division of Genetics and Genomics—who are all professors at Harvard Medical School and associate members of the Broad Institute of MIT and Harvard—say their study findings may provide insights into new Alzheimer’s disease diagnostics and treatments.
“We find that to some extent, Alzheimer’s disease is a little like cancer—driven by the same mutations that drive blood cancers like lymphoma and leukemia,” said Walsh. “This is helpful because we have a lot of drugs to fight cancer and some of them might be useful therapeutically for Alzheimer’s disease.”
The researchers reported on their work in Cell, in a paper titled “Somatic cancer variants enriched in Alzheimer’s disease microglia-like cells drive inflammatory and proliferative states.”
Microglia function as the brain’s resident immune cells, acting as garbage collectors, eating debris and infected or dying cells. “The importance of microglia in Alzheimer’s disease (AD) pathogenesis has been demonstrated by large-scale genetic association studies, which have identified AD risk variants in a growing list of microglia-related genes,” the authors wrote. “Once abnormally reactive in AD, microglia can promote synaptic and neuronal loss while exacerbating tau proteinopathy.”
Unlike the rest of the immune system cells that circulate in the blood throughout the body, microglia don’t cross the blood brain barrier—or so experts thought. For their newly reported study the research team sequenced 149 cancer-driving genes from tissue samples in 190 brains donated from people with Alzheimer’s disease compared to 121 healthy brains. The Alzheimer’s samples had more single DNA letter changes than the healthy tissue with the most changes found repeatedly in the same five cancer driver genes, meaning the microglia were amassing mutations in specific genes. “Deep (>1,000×) panel sequencing of 311 brain samples revealed enrichment of somatic single-nucleotide variants (sSNVs) in cancer driver genes in AD brains, especially in genes associated with clonal hematopoiesis (CH),” the team stated.
The cancer gene mutations the researchers discovered in the microglia are commonly found in blood cancers. Because of this, the team tested blood samples from people with Alzheimer’s disease for these same mutations. The team didn’t expect the blood to have these mutations. However, Walsh’s team found the blood cells of the same Alzheimer’s patients carried the same cancer mutations too.
![Microglia-like immune cells with cancer mutations (purple) emerge in the brain. Separately, clumps of proteins, like Tau or amyloid, accumulate in the brain, making the environment hostile. Those microglia cells with mutations get selected for survival and proliferation, creating an inflammatory environment that makes innocent bystander neurons die, contributing to Alzheimer’s disease. [Christopher Walsh and colleagues at Boston Children's Hospital]](https://www.genengnews.com/wp-content/uploads/2026/04/Low-Res_Walsh-Cell-photo-300x300.jpg)
“These sSNVs were associated with clonal expansion and carried by both microglia-like brain macrophages (MLBMs) in multiple brain regions as well as paired blood, suggesting a likely hematopoietic origin,” the investigators stated. “It was actually a really unexpected finding that suggests a totally new mechanism for Alzheimer’s disease pathogenesis,” said Huang. “The findings mean that the blood’s immune cells with cancer mutations are likely getting into the brain and contributing to disease.”
The researchers theorize that the blood-brain barrier weakens, either by age or injury, allowing the blood’s immune cells to cross into the brain. These new arrivals then convert into microglia-like cells. Separately, clumps of proteins accumulate in the brain, triggering microglia to proliferate and respond. The cells most likely to dominate are those with a selective advantage, such as the microglia-like cells with the cancer mutations. However, these mutant microglia also make the environment more inflammatory and hostile than that of the healthy microglia, causing innocent bystander neurons to die off, which leads to Alzheimer’s disease. “These findings suggest that clonal somatic driver variants in MLBMs are enriched in AD, potentially promoting neuroinflammation and neurodegeneration,” the researchers noted. “Potential roles of somatic cancer driver variants in AD pathogenesis open up a whole new range of therapeutic avenues in AD, complementary to approaches emphasizing amyloid and tau.”
Lee added, “Because it’s hard to access brain tissue in a living patient, genetic screens using blood samples could be developed to test whether a person carries these mutations, and has an increased risk of developing Alzheimer’s disease.” Lee and Huang performed a follow-up study, now posted as a preprint on bioRxiv. Here, they demonstrated that cancer driver mutations observed in patient blood samples increased risk of Alzheimer’s disease independently of a well-established genetic risk factor, APOE4.
The post Alzheimer’s Linked to Cancer Mutations in Brain Immune Cells appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
RFK Jr. says China is ‘eating our lunch’ in biotech advances
China is “eating our lunch” on new drug approvals and clinical trial starts, HHS Secretary Robert F. Kennedy Jr. told Congress Tuesday, while praising the FDA’s actions so far.
“We are losing scientists, we’re losing …
Uncategorized
Tortugas debuts with $106M and brain drugs from Hansoh, Eisai
Led by former Sage Therapeutics executives Jeff Jonas and Al Robichaud, the company is starting out with a portfolio of neurological medicines in clinical testing.
Led by former Sage Therapeutics executives Jeff Jonas and Al Robichaud, the company is starting out with a portfolio of neurological medicines in clinical testing.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars




