Uncategorized
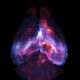
Intercellular Communication via Condensate Corona-Nanoparticle Complexes
Cells and tissues have a multitude of methods for intercellular communication. Nanoscale assemblies that transfer proteins and RNAs between cells are known, but the impacts of external additions or synthetic materials is unclear.
Researchers from the University College of Dublin’s Centre for BioNano Interactions (CBNI) explored detailed changes in nanostructure-biological hybrid complexes as they leave one cell and enter another.
“We had long believed that there are natural couriers and gateways that allow special, very small particulates to communicate in organisms,” said lead author Kenneth Dawson, DPhil, CBNI director.
The team published their work in a paper titled, “Condensate corona–nanoparticle complexes transfer functional biomolecules between cells” in Nature Materials.
In rare instances, a subset of nanoparticles that enter a cell undergo an unexpected transformation, acquiring a coating known as a “condensate corona.” This corona allows for regulated entrance into the cell.
“By gaining access to these natural gateways, it could be possible to ferry ‘toolkits’ of functional biomolecules, for example, extended corrective messages, directly into previously inaccessible areas within cells, and across biological barriers, greatly improving the effectiveness and, importantly, the safety of RNA-, gene- and protein-based therapies,” said lead author associate professor Yan Yan, PhD, UCD School of Biomolecular and Biomedical Science.
Using “magnetic-cored, silica-shelled nanoparticles precoated with a grafted or adsorbed biomolecular corona,” the researchers created a scaffold that provided the cell with a recognition cue, allowing for the cells to deposit a secondary corona. With magnetic cores, and silica shells that carry fluorescent labels, the nanoparticles are easily controlled, extracted, and visualized.
Live-cell imaging showed that these additionally transformed nanoparticles were re-exported and retained both their original corona, along with their new cell-derived layer.
“By combining magnetic core extraction with an optimized pulse–chase regime and post-isolation washing, we obtained highly reproducible particle-complex isolates with minimal background contamination,” the authors wrote. Analysis showed that the cell-derived corona was “solid-like, structurally stable and biochemically robust.”
They also identified protein profiles using stable-isotope amino acid labelling (SILAC) in the cells producing the corona, followed by mass spec analysis. These proteins have a high affinity for the ER and mitochondria and about 70% of the proteins have been previously associated with mesoscopic intracellular RNA granules.
“With the prototype in our hands, we were able to break into these communications and understand how biological information is shared between cells. From there, we began to send our own messages via the same system,” Dawson noted.
In further tests, the team found that within endosomes of the recipient cell, the corona detaches from the core and the fates of the core and corona diverge, with the proteins and RNA components of the corona escaping the endosome—and escaping degradation—to be distributed within and access targets in the cell. They were able to disrupt this process and keep the corona and the attached materials, in the endosome by grafting short peptides onto the coronal surface.
Utilizing CRISPR-Cas9 they tested the functionality of corona-bound particles that escape the endosome. They generated particle complexes for bioluminescent markers to monitor functionality. Analysis revealed “intact enzymatic activity can be delivered to recipient cells by condensate-borne cargo.”
The authors explained that together, their data suggest these condensates function as an encoded biomolecular transfer program that are activated by the recipient cell. They wrote: “It is remarkable that such architectures, built entirely from endogenous biomolecules of producer cells, can embody transfer programs that overcome most of the challenges faced within nanoscale therapeutics.”
“The findings provide a new blueprint for sending strategic and therapeutically effective biological messages to currently inaccessible locations in the body. That points towards a new concept of medicine that could reverse, rather than manage, currently intractable diseases,” concluded Dawson.
The post Intercellular Communication via Condensate Corona-Nanoparticle Complexes appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Brain Astrocytes Form Far-Reaching Connections in Mice
A study in mice headed by NYU Langone Health researchers has found that cells long thought to play a secondary role in brain function build their own far-reaching connections. These pathways appear to connect distant regions in ways that had not been mapped before.
Experts usually describe the brain as a network of nerve cells (neurons) that send each other signals to pass along information. These neurons are maintained by another kind of brain cell, the star-shaped astrocyte, which ferries in nutrients and carries away waste.
The newly reported study, headed by Melissa Cooper, PhD, a postdoctoral fellow in the department of neuroscience at NYU Grossman School of Medicine, revealed that, like neurons, astrocytes form organized webs, which enable them to communicate with other specific astrocytes across the brain rather than only sending local, generalized signals. In some cases, the pathways were found to link areas that were not already joined together by neurons.
“For more than a century, neuroscientists have thought of neurons as the main actors in the brain,” said Cooper. “Yet our findings suggest that astrocytes, which are usually viewed as merely support cells, are also running their own widespread signaling pathway, adding another layer to how brain regions stay connected.” The team suggests that while their study was carried out in mice, not humans, the findings form the basis for future studies investigating how astrocyte networks might link with injury, disease, or aging and to learning and memory.”
Cooper is first and co-corresponding author of the team’s published work in Nature, titled “Astrocytes connect specific brain regions through plastic networks,” in which the researchers stated, “Astrocyte networks can directly link brain regions that are not connected by neurons, suggesting that previously unassociated brain regions communicate with one another through gap junction-coupled astrocytes.”
“Neuronal axons have traditionally been considered to be the primary mediators of functional connectivity among brain regions,” the authors wrote, and the role of communication mediated by astrocytes has been largely underappreciated. “This communication occurs through gap junctions—membrane channels that connect the cytoplasm of neighboring cells, enabling them to redistribute resources and share biochemical signals,” the team continued. “Studies using mice lacking astrocyte gap junctions have shown that these gap junctions are necessary for memory formation, synaptic plasticity, coordination of neuronal signaling, and closing the visual and motor critical periods.”
In earlier work, Cooper reported that in a mouse model of the visual neurodegenerative disease glaucoma, astrocytes can redistribute resources from astrocytes around healthy neurons to damaged neurons. Yet the team had no way to see whether this kind of support-cell network extended across the entire brain.
Cooper said the newly reported study is the first to map active, brain-wide communication networks built by astrocytes and to show that these pathways are highly specific. The research relied on a custom-built tracing tool that let the team follow the cells’ connections in far greater detail than had been possible using past methods. “Despite the importance of astrocyte gap junctional networks, studying them has been challenging,” the investigators noted. “Current methods such as slice electrophysiology disrupt network connectivity and introduce artefacts due to tissue damage.”
For their study, the researchers used a harmless virus to deliver “network tracers” into astrocytes in selected brain regions of lab mice. These tracers tagged small molecules as the molecules passed through the gap junctions linking one astrocyte to another, allowing the team to see which cells were part of the same signaling pathway.
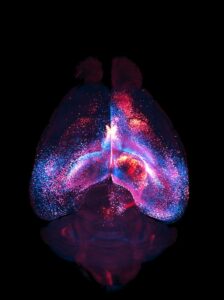
The scientists then made the mice’s brains transparent and used a specialized microscope to capture three-dimensional images of every tagged astrocyte. By doing this across hundreds of mice, they could map astrocyte webs across brain areas. “These networks selectively connect specific regions, rather than diffusing indiscriminately, and vary in size and organization,” they reported. “We observe local networks that are confined to single brain regions and long-range networks that robustly interconnect multiple regions across hemispheres, often exhibiting patterns distinct from known neuronal networks.”
![A 3D network of interconnected astrocytes imaged inside a whole, transparent mouse brain. Each astrocyte's color shows its distance from the viewer; closer astrocytes are blue, while more distant astrocytes are red. [Cooper et al. Astrocytes connect specific brain regions through plastic networks. Nature. 2026. doi:10.1038/s41586-026-10426-6.]](https://www.genengnews.com/wp-content/uploads/2026/04/Low-Res_Cover-Image-224x300.jpg)
The tracing tool and brain-clearing method were designed to be relatively low-cost and easy to reproduce so that other labs could use them to study the networks in many brain diseases.
In another part of the study, the team assessed mice that were genetically engineered with astrocytes that lacked gap junctions. The communication networks largely disappeared, suggesting that the pathways are active and depend on these physical bridges.
“By challenging our understanding of how the brain communicates over long distances, our results may offer fresh insight into how it develops, ages, and behaves in conditions such as Alzheimer’s and Parkinson’s diseases,” said study co-senior author Shane A. Liddelow, PhD, an associate professor in the neuroscience and ophthalmology departments at NYU Grossman School of Medicine.
Another key finding was that astrocyte networks are dynamic. When the team trimmed whiskers on one side of the mice’s faces—“this manipulation is known to induce robust structural remodeling in neurons,” the team noted—a pathway from the region that processes whisker touch got smaller and reconnected to different astrocyte partners.
“The fact that astrocyte networks shrink and reroute after a loss of sensory signals suggests they may be shaped by experience,” said study co-senior author Moses V. Chao, PhD, a professor in the cell biology, neuroscience, and psychiatry departments at NYU Grossman School of Medicine. “It also raises the possibility that each of us has a somewhat unique pattern of connections molded by what our brains have learned and lived through.”
The authors plan to investigate which molecules move through the networks and to apply their tracing tool to models of brain disorders. They also hope to examine how these webs change during development and aging, said Chao.
Liddelow emphasized that while gap junctions and astrocytes exist in humans, it remains unknown whether the networks link the same regions in the same way as in mice. Nevertheless, in their paper, the team concluded that their findings “… establish foundation for future exploration of how astrocyte network structure and function are shaped by injury, disease, development, aging and experience-dependent processes such as learning and memory.”
The post Brain Astrocytes Form Far-Reaching Connections in Mice appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
AbbVie’s North Carolina $1.4B manufacturing site to finish during Trump’s term
AbbVie has provided new details about its $100 billion pledge to the US, budgeting $1.4 billion for its first manufacturing campus in North Carolina.
Construction of the 185-acre campus in Durham, NC, will start this …
Uncategorized
STAT+: At AACR, talk of Chinese biotech, oncology’s comms issue, and more
You’re reading the web version of STAT’s popup newsletter, AACR in 30 seconds, your guide to what’s happening at the American Association of Cancer Researchers’ annual meeting.
This is the last edition of our pop-up newsletter. We hope you’ve learned as much as we have. If you’re not already a STAT+ subscriber, consider it! There’s currently a 60% off promotion on annual subscriptions.
In the meantime, thanks for joining us.
Overcoming resistance and RevMed’s next drug?
In case you missed it, Revolution Medicines’ sessions yesterday were jam-packed with conference attendees. While most of the media coverage focused on the daraxonrasib in frontline pancreatic cancer data, the company also revealed some activity in a new compound, RM-055. CEO Mark Goldsmith described it as being part of a new class of “catalytic inhibitors,” since it can slice off a phosphate from GTP-RAS, or the “on” form of RAS, and turn the protein off.
This generated a lot of interest because one of the main ways that cancer develops resistance to RAS inhibitors is by amplifying mutant RAS, basically flooding the cell with the oncoprotein and overwhelming the inhibitor. RM-055, with its catalytic ability to turn multiple mutant RAS proteins off, may be the next step in the arms race against RAS-addicted cancer.

You’re reading the web version of STAT’s popup newsletter, AACR in 30 seconds, your guide to what’s happening at the American Association of Cancer Researchers’ annual meeting.
This is the last edition of our pop-up newsletter. We hope you’ve learned as much as we have. If you’re not already a STAT+ subscriber, consider it! There’s currently a 60% off promotion on annual subscriptions.
In the meantime, thanks for joining us.
Overcoming resistance and RevMed’s next drug?
In case you missed it, Revolution Medicines’ sessions yesterday were jam-packed with conference attendees. While most of the media coverage focused on the daraxonrasib in frontline pancreatic cancer data, the company also revealed some activity in a new compound, RM-055. CEO Mark Goldsmith described it as being part of a new class of “catalytic inhibitors,” since it can slice off a phosphate from GTP-RAS, or the “on” form of RAS, and turn the protein off.
This generated a lot of interest because one of the main ways that cancer develops resistance to RAS inhibitors is by amplifying mutant RAS, basically flooding the cell with the oncoprotein and overwhelming the inhibitor. RM-055, with its catalytic ability to turn multiple mutant RAS proteins off, may be the next step in the arms race against RAS-addicted cancer.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars