Uncategorized
Hydrogel-Based Axon Model Improves Early Testing for MS Remyelination Therapies
Axons—the long, cable‑like projections that relay electrical signals across the nervous system—depend on tightly wrapped layers of myelin to keep those messages fast and reliable. When this insulation is damaged, as in multiple sclerosis (MS) and other neurodegenerative diseases, signal transmission slows and neurons eventually degenerate. Although oligodendrocytes can repair myelin early on in the process, this capacity declines with age and repeated inflammatory attacks, leaving researchers searching for therapies that can restore myelin more effectively.
A team at University College London (UCL) has now developed a more physiologically realistic way to study how myelin forms—and how potential drugs might influence that process. Their new hydrogel‑based axon model, described in Nature Methods in a paper titled “Tunable hydrogel‑based micropillar arrays for myelination studies,” recreates both the geometry and softness of real axons. The platform is designed to address a longstanding problem in the field: many drug candidates that appear promising in rigid, plastic‑based lab models ultimately fail in human trials.
“To stop MS, we need therapies that repair myelin,” said senior author Emad Moeendarbary, PhD, professor of cell mechanics and mechanobiology at UCL and CEO of BioRecode. “Promising drug candidates in the past have failed when tested in human patients. One factor might be that laboratory models do not replicate the basic physical properties of the human brain.”
The UCL team engineered vertical micropillars—each tens of times thinner than a human hair—using a microfabrication process called photolithography that allowed them to precisely tune diameter, spacing, and stiffness. Unlike earlier artificial axons made from hard polymers, these pillars are composed of polyacrylamide hydrogel, a material whose elasticity can be adjusted to match the ~5 kPa softness of native axons. As the authors noted in the paper, the system “mimics the three‑dimensional architecture and softness of axons,” enabling oligodendrocytes to form “multilayered compact myelin” around the pillars.
The researchers seeded the hydrogel pillars with human and rodent oligodendrocytes and tested several candidate remyelination drugs. When the pillars were tuned to realistic softness, drug performance dropped—suggesting that overly rigid models may have produced misleading hits in the past. “Our work suggests that commonly used rigid models, hundreds of times stiffer than real axons, can generate misleading drug hits,” Moeendarbary said. “We believe that our more life-like model can be used as a more robust early test of drug candidates and as a platform to discover new drugs.”
The study also marks the first demonstration of compact, multilayered myelin grown from human oligodendrocytes in a fully hydrogel‑based system. The platform’s design allows high‑content imaging, transcriptomic profiling, and systematic variation of mechanical cues—capabilities that could help researchers dissect how myelin forms and why it fails in disease.
Building such a soft, microscale structure was not trivial. “Hydrogel is a close mimic of living cells… but to fabricate a soft hydrogel at such a small scale is not an easy task,” Moeendarbary noted, crediting the five years of work led by PhD student Soufian Lasli and Claire Vinel, PhD.
By more faithfully recreating the physical environment of the brain, the UCL team hopes their model will provide a more reliable proving ground for remyelination therapies before they reach clinical trials.
The post Hydrogel-Based Axon Model Improves Early Testing for MS Remyelination Therapies appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Opinion: STAT+: Former Geisinger CEO: U.S. health systems must replace huge numbers of people with AI
About 20 years ago, I stepped on stage at one of our Geisinger town halls and looked out upon a sea of people: thousands of full-time employees at an integrated health system charged with the health and well-being of millions of Pennsylvanians.
Only a fraction of the people in that room were clinicians.
That was the first time I fully visualized the problem: We employed more people in our revenue cycle department to process bills and reconcile data than we did doctors. And we weren’t alone. It’s the same story at every health system in America, large and small, and over the past two decades, the ratio has become dramatically more disparate.

About 20 years ago, I stepped on stage at one of our Geisinger town halls and looked out upon a sea of people: thousands of full-time employees at an integrated health system charged with the health and well-being of millions of Pennsylvanians.
Only a fraction of the people in that room were clinicians.
That was the first time I fully visualized the problem: We employed more people in our revenue cycle department to process bills and reconcile data than we did doctors. And we weren’t alone. It’s the same story at every health system in America, large and small, and over the past two decades, the ratio has become dramatically more disparate.
Uncategorized
A star scientist showed that better genetics lessons could reduce racism. It was the death knell for his career
Every year, the Genetics Society of America bestows the Elizabeth W. Jones Award for Excellence in Education, recognizing someone who has helped the public better understand the science of DNA. It’s understood to be a lifetime achievement award; past recipients tend toward retirement age with decades of work behind them and stacks of textbooks to their names.
When this year’s winner, Brian Donovan, was announced at the end of February, many geneticists and science educators found it hard to celebrate the news. Not because he’s undeserving of the honor. Far from it. But because it seemed to confirm what many feared: that Donovan’s incandescent research career was over before it had barely begun.

Every year, the Genetics Society of America bestows the Elizabeth W. Jones Award for Excellence in Education, recognizing someone who has helped the public better understand the science of DNA. It’s understood to be a lifetime achievement award; past recipients tend toward retirement age with decades of work behind them and stacks of textbooks to their names.
When this year’s winner, Brian Donovan, was announced at the end of February, many geneticists and science educators found it hard to celebrate the news. Not because he’s undeserving of the honor. Far from it. But because it seemed to confirm what many feared: that Donovan’s incandescent research career was over before it had barely begun.
Uncategorized
Microplastics in Human Bile Drive Mitochondrial Dysfunction and Senescence
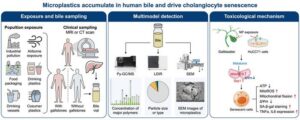
Microplastics have become a defining environmental signature of modern life, turning up in oceans, soil, food, drinking water, and even the air. But their biological fate inside the human body remains far less understood. A new study suggests that these particles may be doing more than simply passing through. Instead, they may be accumulating in one of the body’s most overlooked fluids—bile—and leaving behind measurable cellular damage that could shape future thinking about environmentally driven biliary injury and long‑term health effects. As the authors noted in their abstract, “the long-term accumulation patterns and chronic toxic effects of microplastics within the human biliary system are largely unknown,” underscoring the need for deeper investigation into how these particles behave in the enterohepatic circulation.
Researchers from the Tenth Affiliated Hospital of Southern Medical University (Dongguan People’s Hospital), Sun Yat-sen University, Guilin Medical University, and collaborating institutions reported the findings in Environmental Science and Ecotechnology. Their study, “Microplastics accumulate in human bile and drive cholangiocyte senescence,” provides the first direct evidence that microplastics are not only present in bile but may also contribute to mitochondrial dysfunction and premature aging in cholangiocytes, the epithelial cells that line the bile ducts.
The team collected bile from 14 surgical patients (five without gallstones and nine with gallstones) and used a multimodal analytical approach—pyrolysis–gas chromatography–mass spectrometry, laser direct infrared spectroscopy, and scanning electron microscopy—to characterize the particles. According to the paper, “we show the universal presence of microplastics in human bile,” identifying six polymer types dominated by polyethylene terephthalate and polyethylene, with most particles measuring 20–50 μm. Patients with gallstones carried substantially higher microplastic burdens, raising questions about whether biliary stasis or altered bile composition may influence microplastic retention.

To probe biological effects, the researchers exposed cultured human cholangiocytes to low-dose polystyrene nanoplastics for seven days, simulating chronic exposure. The cells exhibited mitochondrial dysfunction, elevated reactive oxygen species, reduced ATP, Drp1‑mediated mitochondrial fission, and G1 cell‑cycle arrest—hallmarks of senescence. As the authors wrote, chronic exposure “induces mitochondrial dysfunction-associated senescence in cholangiocytes,” suggesting a mechanistic link between environmental microplastics and biliary aging.
One of the most intriguing findings is that melatonin, a widely used antioxidant, partially reversed the mitochondrial and inflammatory damage. While far from a therapeutic recommendation, the result hints at a potential intervention point and gives the study translational relevance.
The work reframes the biliary system as something far more active than a simple transit channel. The data indicate that bile can serve as a reservoir for microplastics and that prolonged exposure may age cholangiocytes by driving mitochondrial dysfunction. The partial rescue with melatonin adds a mechanistic foothold for future intervention, even as the authors caution that broader human studies are essential.
For biotech, the implications are broad. The work highlights bile as a clinically accessible matrix for exposure assessment, opening the door to new diagnostics for environmental toxicology. The mitochondrial stress signature aligns with pathways already being targeted by companies developing senolytics, mitoprotective agents, and anti‑inflammatory therapeutics. The authors wrote that the research provides “a mechanistic foundation for assessing the health risks of plastic pollution and developing therapeutic interventions for environmentally driven biliary disorders.”
The post Microplastics in Human Bile Drive Mitochondrial Dysfunction and Senescence appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors