Uncategorized
Bristol Myers’ pivotal year; Should AI be swinging bigger?; and more
Bristol Myers’ pivotal year; Should AI be swinging bigger?; and more
Uncategorized
Federal appeals court blocks mailing of abortion pill mifepristone
A federal appeals court has restricted access to one of the most common means of abortion in the U.S. by blocking the mailing of mifepristone prescriptions.
Friday’s unanimous ruling from a three-judge panel of the New Orleans-based 5th U.S. Circuit Court of Appeals is requiring that the abortion pill be distributed only in person and at clinics, overruling regulations set by the federal Food and Drug Administration.
Uncategorized
Arvinas’ ‘Protac’ breast cancer drug cleared by FDA
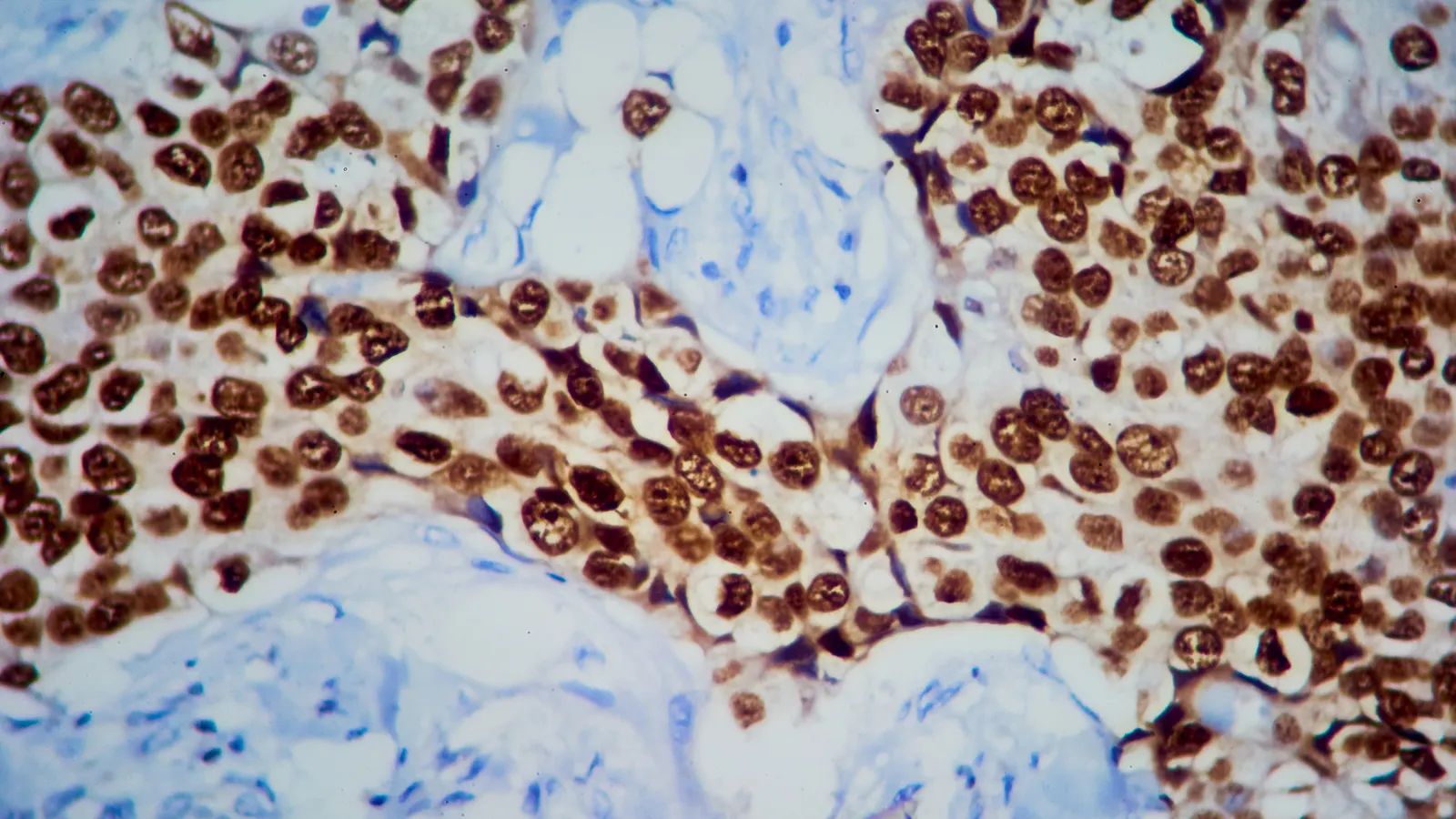
Known as Veppanu, the drug is the first of its kind to get to market. But Arvinas and partner Pfizer are still searching for a new partner to sell it.
Uncategorized
DNA-Containing Extracellular Vesicles Boost Antitumor Responses in Mice
A study led by investigators at Weill Cornell Medicine has found that activated T cells secrete extracellular vesicles (EVs) containing DNA, which can enter other immune and tumor cells to stimulate the body’s defense systems. Preclinical experiments showed that this vesicle-associated DNA could be useful therapeutically, boosting T cell attacks against tumors that otherwise evoke little or no immune response.
Studies in live mice showed that these activated T cell-derived-EVs (AT-EVs) enhanced antigen processing and presentation (APP) in tumor cells and dendritic cells (DCs) across different immunologically cold tumors. The ATEVs also synergized with immune checkpoint inhibitors (ICIs) to trigger antitumor immunity and hold back tumor growth.
The discovery extends the scientific understanding of the immune system, identifies a new strategy for boosting immunity against cancers, and potentially offers a new tool for delivering genetic payloads to other cells. “These findings reveal a natural mechanism for treating immunologically silent tumors and other diseases that stem from insufficient immune surveillance,” said David Lyden, MD, PhD, the Stavros S. Niarchos professor in pediatric cardiology and a member of the Gale and Ira Drukier Institute for Children’s Health and the Sandra and Edward Meyer Cancer Center at Weill Cornell Medicine.
Lyden is co-senior author of the researchers’ published paper in Cancer Cell, titled “Activated T cell extracellular vesicle DNA transfer enhances antigen presentation and anti-tumor immunity,” in which they stated, “We uncover a mechanism whereby activated T cell-derived extracellular vesicles (ATEVs) drive a positive feedback loop that enhances antigen presentation and immune responses in normal physiology and cancer … Notably, ATEVs hold promise as an acellular immunotherapy, restoring APP and synergizing with checkpoint blockade in immunotherapy-refractory tumors.”
Most animal cells secrete extracellular vesicles which can contain cargo including proteins, snippets of DNA, and other molecules. “Extracellular vesicles (EVs) are nanoparticles naturally released by all living cells, containing proteins, lipids, and genetic material, that facilitate intercellular communication,” the investigators wrote.
The Lyden lab in recent years has made seminal discoveries about extracellular vesicles and their functions, finding for example that vesicles secreted by tumor cells can influence the immune system’s anti-tumor response. Their findings, they noted, “… raised the possibility that EVDNA from immune cells, such as T cells, may also have immune-related functions.” For their new study the team examined the roles of vesicles secreted by immune cells, and specifically T cells, which are the immune system’s principal tumor-fighters.
In their initial experiments, the scientists found that under physiological conditions, T cell-secreted vesicles tend to home to lymph nodes, spleen and other centers of immune activity. There the vesicles are preferentially taken up by antigen-presenting immune cells, including dendritic cells, which assist in T cell activation, a critical process in the immune response. The researchers found that the overall effect of these vesicles released by activated T cells is to boost the antigen-presenting process, thus promoting T cell priming and broader immune activation. The key payloads in these immune-boosting vesicles turned out to be snippets of T cell DNA.
“These surprisingly abundant DNA fragments are mostly on the surfaces of the vesicles, and are not just random—they are enriched for immune-related genes, including genes that help cells display antigens to the immune system,” said co-senior author Haiying Zhang, PhD, an assistant professor of cell and developmental biology in pediatrics and member of the Lyden lab. “We also found that these vesicles have, attached to their surfaces, a special enzyme that acts as a molecular drill, enabling the transfer of vesicle-carried DNA into the nucleus of the recipient cell where they can be expressed transiently,” added study co-first author Diao Liu, PhD, a postdoctoral research associate in the Lyden Lab.
Infusing DNA-carrying vesicles from activated T cells into mice with tumors, the researchers found that the vesicles were taken up not only by antigen-presenting cells but also by tumor cells themselves. The treated tumors grew more slowly and were better infiltrated by T cells and other immune cells, indicating that the vesicles induced a stronger anti-tumor response. “Our work reveals an EV-mediated mechanism through which activated T cells enhance APP across diverse recipient cells—from DCs in physiological conditions to cancer cells across tumor types,” the authors noted. Although cancers—and viruses—frequently suppress the antigen-presenting process to make malignant or infected cells “invisible” to the immune system, the main effect of the extracellular vesicular DNA was to reverse this process, restoring tumor cells’ visibility.
The team demonstrated the effectiveness of this approach, alone and in combination with existing immunotherapy, in preclinical models of three different immunologically silent cancers: glioblastoma, pancreatic and triple-negative breast cancer. “By boosting APP machinery, ATEVs enhance tumor immunogenicity and elicit robust anti-tumor responses, particularly when combined with ICIs in otherwise resistant tumors, including pancreatic, breast, and brain cancers,” they stated. “These findings reveal the translational potential of activated T cell-derived extracellular vesicles (ATEVs) by exploiting a naturally occurring immune-boosting process to overcome immune evasion, particularly in immunologically silent cancers.”
Co-senior author Irina Matei, PhD, an assistant professor of immunology research in pediatrics and member of the Lyden lab, stated, “There seems to be a positive-feedback loop, in which the DNA-carrying vesicles from activated T cells amplify the immune response by acting on both antigen-presenting cells, which increase expression of the machinery processing tumor antigens, and tumor cells, promoting their recognition by the immune system as well as their own production of DNA-laden vesicles.”
The researchers are now working to translate their findings into a new, vesicle-based cancer treatment, which could be used on its own or in conjunction with standard immunotherapies or other cancer treatments. “The surprising ability of these vesicles to transfer DNA from donor T cells into the nuclei of recipient cells suggests their potential as a natural, non-viral platform for transient gene delivery,” said co-first author Mengying Hu, PhD, a postdoctoral research associate in the Lyden Lab who led the research and is now an assistant professor of pharmaceutical sciences at the Ohio State University. “The results point to a broadly applicable gene-transfer strategy that may offer improved safety and efficiency compared with current gene therapy approaches.”
In their paper the authors concluded, “Overall, ATEVs emerge as an acellular immunotherapy and delivery modality that can prime antitumor immunity, synergize with existing therapies, and serve as a vaccine adjuvant,” they concluded. “Our findings provide a foundation for the therapeutic application of ATEVs through a deeper understanding of the biological role of AT-EVDNA.”
The post DNA-Containing Extracellular Vesicles Boost Antitumor Responses in Mice appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars