GEN – Genetic Engineering & Biotechnology News
After Buying HUB and Partnering with Promega, MilliporeSigma Charts Growth Path in Organoids
Drug developers and their contract development and manufacturing organizations (CDMOs) moving away from animal testing—whether for financial, regulatory, or moral reasons—are increasingly finding an alternative in organoid models designed to uncover biological insights previously unattainable through traditional cell culture experiments or animal models.
Merck KGaA, Darmstadt, Germany, and its U.S. and Canada Life Science business, MilliporeSigma, set out to establish a leading presence in organoid development last year when it completed its €104 million ($120 million) acquisition of HUB Organoids. In the 14 months that followed, MilliporeSigma and its parent company have expanded their footprint in organoids, through internal efforts and external partnerships.
Those began with the purchase of HUB Organoids. Headquartered in Utrecht, the Netherlands, HUB holds a foundational patent portfolio focused on organoids, and offers services that include generating new models, developing assays, and high-throughput screening.
“What we see, driven by our customers and the need in biology, is that the 2D cell culture is not enough to predict what actually happens in humans,” Karen Madden, PhD, senior vice president and chief technology officer with MilliporeSigma, told GEN. “People have been wanting to move to more sophisticated models like 3D cell culture, and now ultimately organoids of different types, and ideally organoids that are derived from human tissue, because that’s even more predictive.”
Madden said Merck KGaA’s acquisition of HUB Organoids “gave us access to the world’s leading and proprietary position around these human-derived, or patient-derived organoids. So, we’re commercializing those, scaling them up, we’re adding automation to them.”
‘Growing from scratch’
HUB is short for “Hubrecht Organoid Technology,” reflecting the company’s spinout after being founded in 2013 by the Hubrecht Institute, UMC Utrecht, and the Royal Netherlands Academy of Arts and Sciences (KNAW). “We set out to develop the technology and we always said, we want to be the market leader in this organoid field,” HUB CEO Robert Vries, PhD, said on a video posted last year to mark the company’s 10th anniversary. “As a company coming from scratch, it’s really fantastic to see its [the technology’s] adoption, and that you can play a major role in a field like this.”
Adoption of organoid technology comes as biopharma drug developers, universities, and research institutions have been shifting away from animal testing toward new approach methodologies (NAMs) such as organoids as well as other 3D models such as spheroids and organs on chips. Other examples of NAMs include complex and two-dimensional in vitro studies, chemical reactivity studies, computer simulations or in silico modeling, and studies based on phylogenetically lower animals such as zebrafish or C. elegans.
The FDA has sought to advance NAMs through the FDA Modernization Act 2.0 enacted in 2022, which removed the animal testing requirement for new FDA-regulated products that was imposed through the Federal Food, Drug, and Cosmetics Act of 1938. As part of the agency’s Roadmap to Reducing Animal Testing in Preclinical Safety Studies, released in April 2025, the FDA just on Wednesday issued a draft guidance designed to help drug developers validate new NAMs.
“As FDA gains confidence in these tools, they could be formally adopted to reduce or replace specific animal tests,” according to the 11-page draft guidance.
In October, the FDA approved the first-ever investigational new drug (IND) submission supported solely through human vascularized organoid–based combination studies, without relying on traditional animal efficacy (POC) testing.
The IND application by SillaJen enabled the South Korea-based developer of oncolytic virus immunotherapeutics to begin clinical trials for a combination therapy consisting of tislelizumab or paclitaxel and BAL0891, a dual inhibitor of Threonine Tyrosine Kinase (TTK) and polo-like kinase 1 (PLK). SillaJen’s combo therapy incorporating BAL0891 is being evaluated in a Phase I trial (NCT05768932) whose primary completion date is estimated at December 24. SillaJen’s IND included preclinical efficacy data generated through the vascularized tumor immune microenvironment model (vTIME) developed by Qureator.
Business integration
Soon after acquiring HUB Organoids, Merck KGaA initially integrated the business into the former Science and Lab Solutions (SLS) unit of its Life Sciences business segment, which includes tools, services, and laboratory equipment for pharmaceutical R&D and manufacturing. That unit is also poised for growth once Merck KGaA completes its acquisition of the chromatography business of JSR Life Sciences, a closing expected by the end of the second quarter.
Last year, the Life Sciences segment finished with €8.98 billion ($10.36 billion) in revenue. Merck KGaA does not break down its businesses further than its three segments, which also include healthcare (drug development, focused on oncology, neurology and immunology, and “global health” treatments such as for malaria) and electronics (high-tech materials).
But in a restructuring that took effect at the start of this year, Merck KGaA broke up SLS into two units, Advanced Solutions and Discovery Solutions—with HUB Organoids and organoid activity included within Discovery Solutions.
Last year, the businesses that comprise Discovery Solutions generated €2.829 billion ($3.26 billion) in sales, with quarter-over-quarter “organic” or non-acquisition growth ranging from -4% to +3%. Fourth quarter 2025 sales were negatively impacted by the U.S. federal government shutdown and funding uncertainties, Merck KGaA told analysts March 5 in its Q4 2025 earnings presentation.
In October, MilliporeSigma and Promega launched a partnership to co-develop new technologies designed to advance drug screening and discovery—specifically, by developing assays capable of tracking cellular activity in real time using a reporter system within organoids that mimic human biology, allowing testing in models that are physiologically more relevant than traditional two-dimensional (2D) models.
The collaboration intended to marry MilliporeSigma’s strengths in organoids and synthetic chemistry with Promega’s leading assay and reporter technologies.
Reading out organoids
“One important question with these organoids is, how do you read them out after you grow them?” Madden observed. “One way you can read them out is by using really sophisticated cell-based assays that go along with the organoids. Promega is world-renowned for these very novel cell-based assay types that they have. We’re partnering with them to provide a very intricate, very specific, very informative readout to these organoid models that we have, so that we can bring those joint solutions to our customers.”
The organoids are being designed to address several challenges in drug screening. One is the need to track cellular activity. Other challenges relate to speed and efficiency.
“Some of the challenges that we see with our customers and specifically related to organoids is that they want to go very quickly at a very low price with high reproducibility and super high quality from sample, whatever sample that is, to answer or data or readouts,” Madden said. “We’re trying to help solve that by providing these very high quality, reproducible, unique, complex materials, but at a very high quality and very reproducible, trying to add automation and automated solutions to that. And you’ll see that across our entire portfolio.”
MilliporeSigma and Promega are working on both organoids that HUB already has developed, as well as new ones.
“We’re constantly kind of building out the different types of organoids that are tied to specific, anonymized patients, but with the whole clinical record showing other clinical factors and then ultimately the outcomes of these patients, which makes these models very valuable and more predictive of what’s actually going to happen in a human being when you test your model,” Madden said.
The HUB Organoids acquisition and Promega partnership are efforts that are part of what MilliporeSigma and parent Merck KGaA describe as their commitment to providing next-generation biology solutions.
Automation collaboration
To that end, MilliporeSigma early last year signed a multi-year agreement of undisclosed value with lab automation and accessible robotics provider Opentrons Labworks to automate assay kits on a custom Opentrons Flex® workstation. The custom workstation and automation-enabled assays are intended to reduce manual processing and repetitive tasks with a user-friendly robotic system, thus delivering increased consistency and higher throughput.
“We’re putting a lot of our portfolio into these automated systems again, so our customers can do more with less, and it’s a lot more reproducible, and it’s a much higher throughput,” Madden said.
In May, Merck KGaA announced a strategic partnership of undisclosed value with imec, a Belgian-based research and innovation hub in nanoelectronics and digital technologies, aimed at developing an advanced microphysiological systems (MPS) platform that will reduce drug developers’ reliance on animal testing by increasing the predictive validity of next-generation preclinical models.
“By integrating industry leading induced pluripotent stem cells and patient derived organoids portfolio offered by our company with the disruptive joint hardware platform that features an unprecedented number of biosensors, we’re creating a connected in vitro and in silico pipeline capable of generating the high-quality biological training data that is critically needed,” Steven Johnston, vice president and head of technology enablement at Merck KGaA, Darmstadt, Germany, said in a statement.
The post After Buying HUB and Partnering with Promega, MilliporeSigma Charts Growth Path in Organoids appeared first on GEN – Genetic Engineering and Biotechnology News.

GEN – Genetic Engineering & Biotechnology News
Bioengineered Implants Deliver Multi-Drug Therapy in Animal Models
In a new paper, scientists from Northwestern University and their collaborators at Rice University and Carnegie Mellon University report on their progress towards developing so-called implantable “living pharmacies.” These are tiny devices containing engineered cells that continuously produce medicines inside the body. Details of the study, which was done in rats, are published in Device in a paper titled “Design of a wireless, fully implantable platform for in-situ oxygenation of encapsulated cell therapies.”
The device, which is called the hybrid oxygenation bioelectronics system for implanted therapy or HOBIT, is roughly the size of a folded stick of gum. It integrates engineered cells with oxygen-producing bioelectronics and is designed in such a way that the cells are shielded from the body’s immune system while also receiving oxygen and nutrients needed to keep them alive and producing drugs for several weeks. In the future, these devices could be deployed to treat chronic conditions without requiring patients to carry, inject, or remember to take medications.
“This work highlights the broad potential of a fully integrated biohybrid platform for treating disease,” said Jonathan Rivnay, PhD, a professor of biomedical engineering and materials science and engineering at Northwestern and a co-principal investigator of the project. “Traditional biologic drugs often have very different half-lives, so maintaining stable levels of multiple therapies can be challenging. Because our implanted ‘cell factories’ continuously produce these biologics, keeping the cells alive with our oxygenation technology allows us to sustain steady levels [of] multiple different therapeutics at once.”
Solving the oxygenation challenge was critical to the success of HOBIT. When engineered cells are packed together in an implant, they compete for oxygen to live. Without a steady supply, many cells die, which limits how much medicine the implants can produce. In an earlier study, Rivnay and his collaborators demonstrated how a tiny electrochemical device could generate oxygen by splitting nearby water molecules, and showed that supplying oxygen locally dramatically improved the survival of implanted therapeutic cells. The latest iteration of their device integrates that oxygen-generation technology in a fully implantable, wireless system.
Digging into the details of the device, HOBIT contains three primary components: a cell chamber that holds the genetically engineered cells, a miniature oxygen generator, and electronics and a battery to regulate oxygen production and wirelessly communicate with external devices. Because the device produces oxygen directly inside the implant, the cells receive a steady supply even in hypoxic environments. “We are producing oxygen directly where the cells need it,” Rivnay said. “That allows us to support much higher cell densities in a much smaller space.” In fact, “cell densities in HOBIT were roughly six times higher than conventional unoxygenated encapsulation approaches.”
According to the paper, the team engineered the cells to produce three different biologics—an anti-HIV antibody, a GLP-1-like peptide used to treat type 2 diabetes, and leptin, a hormone that regulates appetite and metabolism. They implanted the devices under the skin of rats and monitored drug levels in their bloodstreams for 30 days. Blood measurements of animals with the implanted devices showed sustained levels of all three biologics throughout the study period. In contrast, in animals that were implanted with devices without oxygenation, the biologics that had shorter half-lives were undetectable by the seventh day. Drugs with longer half-lives in these animals also declined steadily over time. At the end of the testing period, roughly 65% of the cells in the oxygenated devices remained viable compared with roughly 20% in control devices.
For their next steps, the scientists intend to test their devices in larger animal models and explore disease-specific applications, including therapies based on transplanted pancreatic cells. “As these technologies continue to develop, devices like this could eventually act as programmable drug factories inside the body—delivering complex therapies in ways that simply aren’t possible today,” Rivnay said.
The post Bioengineered Implants Deliver Multi-Drug Therapy in Animal Models appeared first on GEN – Genetic Engineering and Biotechnology News.
GEN – Genetic Engineering & Biotechnology News
Gut-Immune Link Identified in Multiple Sclerosis-Related Neuroinflammation
Multiple sclerosis (MS) is a debilitating neurological disorder caused by malfunctioning immune responses that target the brain and spinal cord of the central nervous system (CNS). New research led by Shohei Suzuki, MD, PhD, assistant professor, division of gastroenterology and hepatology, and Tomohisa Sujino, PhD, associate professor, School of Medicine, at Keio University, Japan, has now indicated how the gut can initiate neuroinflammation in multiple sclerosis.
Their study found that intestinal epithelial cells (IECs) promote the development of pathogenic T cells that migrated to the spinal cord and induced disease symptoms in mouse models of the disorder.
The researchers examined intestinal tissues from patients with MS and mice with experimental autoimmune encephalomyelitis (EAE), a close analog of MS. In both cases, they observed an increase in TH17 cells and an upregulation of major histocompatibility complex class II (MHC II) expression in IECs. Deleting MHC II in IECs reduced the accumulation of TH17 cells in the gut and lowered the severity of EAE. They suggest the results could inform future strategies for developing targeted therapeutics against autoimmunity.
“While current therapies for MS often target B cells, our study highlights the gut as an important therapeutic site,” Suzuki commented. “Modulating intestinal microbiota or antigen-presenting activity of IECs represents new approaches to treating autoimmune neurological diseases.”
Suzuki, Sujino, and colleagues reported on their findings in Science Immunology, in a paper titled “Intestinal Epithelial MHC Class II Induces Encephalitogenic CD4⁺ T Cells and Initiates Central Nerves System Autoimmunity,” in which they concluded, “Our findings reveal an interaction between gut IECs and neuroinflammatory diseases through MHC II expression in human MS and mouse EAE, providing a mechanistic link between gut immune education and CNS autoimmunity and opening new avenues for targeting intestinal immunity in neuroinflammatory diseases.
Failure of the immune system to distinguish ‘self’ from ‘non-self’ entities leads to excessive autoimmune responses against self-proteins like myelin, which forms a protective covering on the neurons. Multiple factors influence the onset and progression of MS, including genetic susceptibility, environmental triggers, and, more recently, the gut microenvironment. Patients with MS exhibit alterations in their gut microbiota, while the gut microbiota and microbial metabolites play a pivotal role in shaping the chronic autoreactive immune responses. “… in an experimental autoimmune encephalomyelitis (EAE) model, commensal or specific microbes were found to be essential for disease initiation and progression,” the authors wrote.
However, in trying to define this gut–CNS axis, the cellular mechanisms that relay the gut-derived signals to the immune system to influence autoimmune inflammation in the CNS remain poorly understood. “Increasing evidence shows that the gut microbiota influences neurological diseases such as Parkinson’s, Alzheimer’s, and MS,” Sujino stated. “However, the mechanisms linking gut microbes, intestinal immunity, and brain inflammation remain unclear. We were keen to identify how gut immune responses contribute to neuroinflammatory diseases.”
Prior research has shown that gut-derived signals can promote the differentiation of T cells into pathogenic T helper 17 (TH17) in mouse models of MS. Recent studies have suggested that IECs can function as antigen presenting cells that help induce these pathogenic cells, but the underlying mechanisms have been unclear.
Building on their previous observation that mild intestinal (ileal) inflammation exists in experimental autoimmune encephalomyelitis (EAE), which is a mouse model of MS, the authors set out to test whether similar inflammation is present in patients with MS. By performing single-cell RNA sequencing on intestinal biopsies, the team identified that inflammatory Th17 cells accumulate in the mouse EAE model as well as in the intestine of patients with MS, suggesting a conserved gut–CNS axis that may be active in human diseases.
In both EAE mice and patients with MS, intestinal epithelial cells upregulated antigen presentation pathways. Particularly, epithelial cells in the ileum had higher expression of major histocompatibility complex class II (MHC II) that presents antigens to CD4+ T cells. “Clinically, patients with MS exhibited an increased expression of epithelial MHC II–associated genes and an accumulation of CD4 T cells in the small intestine, suggesting the conservation of this gut-CNS axis in human diseases,” the scientists stated. Experiments showed that selective deletion of MHC II in IECs reduced pathogenic Th17 cell generation and disease severity. “Conditional deletion of MHC II in IECs showed that epithelial antigen presentation was indispensable for the local expansion of pathogenic Th17 cells in the gut and their subsequent migration to the CNS,” the team stated.
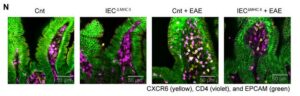
![Immunofluorescence analysis was performed on terminal ileum samples from Cnt, IECΔMHCII, Cnt + EAE, and IECΔMHCII + EAE mice. A total of 3–5 tissue sections were analyzed per mouse, with 3 mice included in each group. [Shohei Suzuki]](https://www.genengnews.com/wp-content/uploads/2026/03/low-res-2-1-300x96.jpeg)
mouse, with three mice included in each group. [Shohei Suzuki]
IECs do not typically present antigens to immune cells. So, the team conducted co-culture assays to test the antigen presentation function of IECs. Their findings demonstrate that IECs can directly present antigens in an MHC II-dependent manner to prime CD4+ T cells in the gut. Notably, in these assays, IECs induced Th17 polarization of activated CD4+ T cells. It became clear that the gut was a critical site for immune activation of pathogenic CD4+ T cells that polarized into pro-inflammatory Th17 cells. “These findings provide direct functional evidence that IEC-expressed MHC II is sufficient to drive Th17 polarization from primed CD4 T cells in an antigen-dependent manner, supporting a direct role for IECs as non-professional antigen-presenting cells,” the scientists reported.
To investigate whether the Th17 cells directly contribute to the pool of autoreactive cells in the CNS, they used transgenic mice that express the Kaede protein, which undergoes photoconversion from green to red fluorescence upon exposure to violet light. This model allowed for precise tracking of pathogenic Th17 cells induced in the intestinal lamina propria that then migrate to the spinal cord and drive neuroinflammation.
Taken together, the study findings reveal a critical role for MHC II expressed by IECs in the expansion of pathogenic Th17 cells that subsequently migrate to the CNS during EAE, providing a mechanistic link between gut immune responses and autoimmune neuroinflammatory diseases. The results demonstrate that while systemic circulation allows T cell exchange across immune tissues, the epithelial–immune interactions within the gut mucosal compartment can essentially shape effector T cell responses in the brain.
“This study reveals a previously unknown role of IECs in antigen presentation and Th17 programming, thereby defining a gut-CNS immunological axis with important implications for understanding and treating autoimmune neuroinflammation,” the authors concluded. “Our findings suggest that the modulation of epithelial antigen presentation could serve as a novel therapeutic approach for MS and related diseases. Given the accessibility of the gut epithelium to dietary, microbial, and pharmacological interventions, targeting IEC–T cell interactions may offer a tractable strategy for immunomodulation.”
The post Gut-Immune Link Identified in Multiple Sclerosis-Related Neuroinflammation appeared first on GEN – Genetic Engineering and Biotechnology News.
GEN – Genetic Engineering & Biotechnology News
Agentic AI, Virtual Cell, LNP Vaccine Boosters, Engineered Organs, and Mergers
This week, agentic AI steps into the limelight buoyed by the momentum from generative AI. And there’s a new virtual cell model in town courtesy of AI-drug developer Xaira Therapeutics. From the frontiers of AI, our discussion turned to feats of engineering in regenerative medicine and lipid nanoparticles. In one study, scientists redesigned LNPs to avoid the liver and accumulate in the lymph nodes. In the other, efforts to develop and implant a lab grown esophagus from donor pigs bear fruit. Finally, Novartis plans to spend up to $3 billion to expand its cancer pipeline with the acquisition of Pikavation Therapeutics. And Merck is acquiring Terns Pharmaceuticals for approximately $6.7 billion also with an eye towards boosting its cancer portfolio.
Listed below are links to the GEN stories referenced in this episode of Touching Base:
NVIDIA GTC 2026: Agentic AI Inflection Hits Healthcare and Life Sciences
By Fay Lin, PhD, GEN Edge, March 18, 2026
Xaira’s First Virtual Cell Model Is Largest To-Date, Toward Complex Biology
By Fay Lin, PhD, GEN Edge, March 25, 2026
Modified Lipid Nanoparticles Boost mRNA Vaccine Delivery to Lymph Nodes
GEN, March 24, 2026
Engineered Esophagus Rebuilds Missing Organ Segment in Pig Models
GEN, March 20, 2026
Novartis Acquires Pikavation for Up to $3B, Expanding Cancer Pipeline
GEN, March 22, 2026
Merck Bolsters Cancer Pipeline with $6.7B Terns Buyout
By Alex Philippidis, GEN Edge, March 25, 2026
Touching Base Podcast
Hosted by Corinna Singleman, PhD
Behind the Breakthroughs
Hosted by Jonathan D. Grinstein, PhD
The post Agentic AI, Virtual Cell, LNP Vaccine Boosters, Engineered Organs, and Mergers appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors



