Uncategorized
Autoimmune Disease-Related Inflammation Reduced with ENDOtollins Drug
A new study published in Nature Chemical Biology titled, “Munc13-4–STX7 inhibitors impair endosomal TLR activation and systemic inflammation,” scientists from Scripps Research have developed a new class of drug compounds, called ENDOtollins, that reduce harmful inflammation while maintaining the body’s ability to fight infections. The results offer new directions to treat autoimmune diseases, such as lupus, and rheumatoid and juvenile arthritis, which together affect more than 15 million Americans.
“A key component of our approach is to begin by understanding the biological mechanisms at play,” said Sergio Catz, PhD, professor at Scripps Research and corresponding author of the study. “By accomplishing this first, we can more easily target the pathway driving inflammation without affecting other important processes.”
Current autoimmune disease treatments, such as hydroxychloroquine, function by broadly blocking endosomes. While effective, this approach can lead to significant side effects, including gastrointestinal problems and, less commonly, vision damage, that cause patients to stop treatment.
The authors focused on two proteins, Munc13-4 and syntaxin 7, that bind together to activate Toll-like receptors (TLRs), immune sensors that activate endosomes. This mechanism plays a key role in detecting foreign DNA and RNA from viruses and bacteria. In autoimmune diseases, TLRs become overactive and trigger chronic, damaging inflammation in the absence of a threat.
The team screened roughly 32,000 compounds and identified molecules that specifically block the Munc13-4–syntaxin 7 interaction without disrupting other cellular functions. Given that Munc13-4 is found mainly in immune cells, the compounds offer a targeted approach to reduce inflammation.
“Most treatments for autoimmune diseases manage symptoms; they don’t change the underlying course of the disease,” said Hugh Rosen, MD, PhD, professor at Scripps Research and co-author of the study. “What’s exciting about this approach is its potential to be disease-modifying: targeting the specific molecular machinery that drives inflammation, rather than broadly suppressing the immune system.”
Notably, the study screened compounds in an intact cellular environment which contrasts from many drug screening approaches, which extract proteins from the cell.
“By maintaining the proteins in their natural environment, we increase the likelihood that compounds we find will actually work in living cells,” said Jennifer Johnson, PhD, first author and senior staff scientist at Scripps Research.
The most potent compound, ENDO12, reduced inflammation in animal models that were also given a TLR-activating molecule. Blood levels of inflammatory markers, including immune system activators IL-6 and IFN-γ, and the enzyme myeloperoxidase, dropped significantly in animals that were treated.
ENDO12 treated animals demonstrated normal antiviral immune response when exposed to a virus. This selectivity addresses the concern that dampening inflammation with immunosuppressive drugs may leave patients vulnerable to infections.
Looking ahead, the team will test ENDOtollins in models that more closely mimic human autoimmune diseases and evaluate the compounds’ chemistry for potential clinical use.
Beyond autoimmune conditions, the researchers suggest ENDOtollins might help treat cytokine storms, the dangerous immune overreactions seen in patients with severe COVID-19 and as a side effect of CAR T cancer therapy. Both involve excessive IL-6 and runaway inflammation.
While translating these findings into treatments for patients remains a long-term goal, Catz emphasizes that the mechanistic insights are valuable in their own right. ENDOtollins can serve as precision tools to probe other cellular processes regulated by endosomes and lysosomes, including pathways implicated in neurodegeneration and immune dysfunction.
The post Autoimmune Disease-Related Inflammation Reduced with ENDOtollins Drug appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
White House adds AbbVie’s Humira to TrumpRx alongside cheaper biosimilars
White House adds AbbVie’s Humira to TrumpRx alongside cheaper biosimilars
The White House this week began offering a $950-per-month version of AbbVie’s megablockbuster Humira on TrumpRx, even as the drug discount website also offers two Humira biosimilars from Pfizer and Amgen that both cost less … Read More
Uncategorized
High-Throughput Platform for Fast-Acting Covalent Protein Therapies
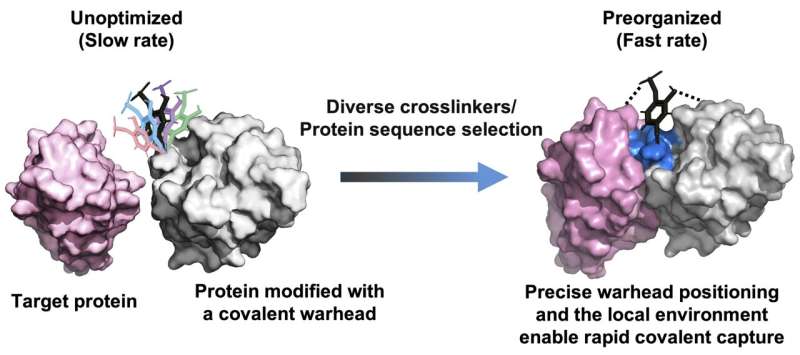
Researchers at Westlake University in China, lead by Bobo Dang, PhD, and Ting Zhou, PhD, report the development of a high-throughput platform for engineering fast-acting covalent protein therapeutics. The team says their study “A high-throughput selection system for fast-acting covalent protein drugs,” published in Science, opens new avenues for next-generation biologics.
Covalent small-molecule drugs have shown great success in cancer therapy by forming irreversible bonds with their targets. This has inspired efforts to extend covalent strategies to protein therapeutics, especially engineered miniproteins. However, their development is limited by a kinetic mismatch. Miniproteins are rapidly cleared in vivo, while covalent bond formation is typically slow. In addition, high-throughput platforms for systematically optimizing covalent protein reactivity have been lacking.
To address this challenge, the researchers proposed that precise spatial positioning of chemical warheads within protein scaffolds could enable molecular preorganization, thereby accelerating covalent bond formation without increasing intrinsic reactivity (see figure).
![The principle for developing fast-acting covalent proteins via comprehensive crosslinker and protein sequence engineering. [Bobo Dang's Lab at Westlake University]](https://www.genengnews.com/wp-content/uploads/2026/04/fast-acting-covalent-p.jpg)
Based on this concept, the team created a high-throughput platform that combines yeast surface display with chemoselective protein modification to screen diverse crosslinkers and millions of protein variants. The platform enables rapid and irreversible target engagement.
Using this platform, the researchers developed a covalent antagonist targeting PD-L1, termed IB101. Structural analysis revealed that IB101 forms a defined binding pocket that precisely positions the active moiety in a reactive conformation, greatly accelerating covalent bond formation.
Functionally, IB101 effectively blocks the PD-1/PD-L1 immune checkpoint pathway and demonstrates strong antitumor activity in mouse models. Notably, despite its short in vivo half-life, IB101 achieves durable target engagement and tumor suppression, outperforming conventional antibody-based therapies under comparable conditions, according to the scientists.
The platform was further applied to cytokine engineering, leading to the development of a covalent IL-18 variant, IB201. This engineered cytokine rapidly forms a covalent interaction with its receptor, enhancing signaling strength and duration. In vivo studies showed that IB201 induces potent antitumor immune responses without detectable systemic toxicity. These results highlight the potential of covalent engineering to improve the efficacy and safety of cytokine-based therapies.
Beyond immunotherapy targets, the platform was also applied to develop a covalent inhibitor targeting the receptor-binding domain (RBD) of SARS-CoV-2. This molecule showed durable viral neutralization, demonstrating the versatility of the approach across different therapeutic modalities, note the researchers, adding that the study establishes a general strategy for engineering fast-acting covalent protein therapeutics.
By enabling covalent bond formation on timescales compatible with rapid in vivo clearance, the platform overcomes a fundamental limitation in the field, say the scientists. These findings, they continue, provide a new framework for designing biologics with both rapid kinetics and sustained target engagement, with broad implications for cancer immunotherapy, antiviral therapy, and beyond.
The post High-Throughput Platform for Fast-Acting Covalent Protein Therapies appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Terns rebuffed a higher bid before selling to Merck

Regulatory filings show that Terns ultimately accepted an offer 15% lower than a previous proposal as four companies vied for rights to the coveted leukemia drug developer.


Regulatory filings show that Terns ultimately accepted an offer 15% lower than a previous proposal as four companies vied for rights to the coveted leukemia drug developer.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors