GEN – Genetic Engineering & Biotechnology News
StockWatch: CytomX Shares Yo-Yo on Colorectal Cancer Data
CytomX Therapeutics (NASDAQ: CTMX) drew attention a few days ago after its six-year-old, up-to-$1.68 billion cancer collaboration with Astellas Pharma (Tokyo: 4503) was terminated by the Japanese pharma giant, but the setback hardly registered with investors of the South San Francisco, CA, biotech.
Instead, what drew investors to CytomX stock on March 16 was its announcement of positive dose expansion data from a Phase I trial (NCT06265688) assessing varsetatug masetecan (varseta-M) in metastatic colorectal cancer (mCRC). Varseta-M achieved an overall response rate (ORR) of 32% and a median progression-free survival (mPFS) of 7.1 months at the high dose of 10 mg/kg.
“We continue to be highly encouraged by what we’re seeing, and we aim to develop varseta-M aggressively for the benefit of patients with CRC and over time, many other cancers,” Sean A. McCarthy, D.Phil., CytomX’s CEO and chairman, told analysts on the company’s quarterly earnings call on March 16.
CytomX basked in the glow of that news as its shares rocketed 75%, soaring from $4.68 to $8.20 early in trading before settling for a 44% gain and a closing price that day of $6.75. But the stock went downhill the following two days, as CytomX shares tumbled 19% to $5.45 Tuesday, then skidded another 19% Wednesday to $4.40.
The 35% nosedive was reversed on Thursday when CytomX shares rebounded 9% to $4.78. But the company gave up most of that gain Friday, through a 7% drop that sent the stock finishing the week at $4.45—down 5% from a week earlier.
Varseta-M is a Probody-developed masked, conditionally activated antibody–drug conjugate (ADC) armed with a topoisomerase-1 inhibitor payload (licensed from drug co-discoverer ImmunoGen, acquired by AbbVie for $10.1 billion) and directed toward epithelial cell adhesion molecule (EpCAM).
“We see a buying opportunity here with a rare ADC asset capable of generating an ~$1B+ opportunity in 3L+ (third- and subsequent line) mCRC alone, with potential ~5B+ as into 1/2L (first- and second-line), and possible expansion into a wide range of solid tumors with high EpCAM expression,” Jefferies equity analyst Roger Song, MD, predicted in a research note.
Examples of applicable solid tumors with high EpCAM expression, Song added, include gastric, ovarian, lung, and breast cancers. Within mCRC alone, Jefferies forecasts a combined ~$2 billion in peak-year adjusted U.S. sales.
Why the selloff?
But if varseta-M is such a potential blockbuster, why then did enough investors sell off their shares to send the stock plunging after the initial surge?
One explanation is CytomX pricing a $250 million public offering of stock and warrants a day after the positive data release, with net proceeds intended for the continued development of varseta-M and other pipeline programs.
CytomX offered 45,990,567 shares of its common stock at $5.30 per share, plus pre-funded warrants allowing selected investors to purchase 1,179,245 shares. The company also granted underwriters of the offering a 30-day option to purchase up to an additional 7,075,471 shares at the public offering price, less underwriting discounts and commissions.
Arguably, a more significant reason was the relatively small number of patients from which the positive data were collected—just 56 evaluable patients, of which 17 were dosed at 7.2 mg/kg, 20 at 8.6 mg/kg, and 19 at 10 mg/kg. At the two lower dosages, CytomX reported ORRs of 6% and 20%, respectively, significantly lower than the 32% the company focused on in its announcement. mPFS durations at the lower dosages were 5.5 months and 6.8 months, respectively, compared with 7.1 months at the high dose.
That 7.1-month figure reflected dose expansion patients who did not receive mandatory prophylaxis and who, according to the company, frequently required early dose interruptions or reductions.
As a result, Song wrote, “it is reasonable to believe” that the dose optimization patient cohort “has materially longer time-on-drug potential” as a result of a change to the prophylaxis regimen that included dosing patients at adjusted ideal body weight starting in Q4 2025 with updated prophylaxis anti-motility medication (loperamide or diphenoxylate/atropine) plus budesonide.
“We think mgmt. [management] clarity and further data disclosure could address investors’ debate/curiosity, mostly driven by smaller N [number of patients]/short follow-up, particularly at dose optimization,” Song commented.
Song’s concerns did not stop him from doubling his firm’s 12-month price target on CytomX shares, from $8 to $16. Jefferies was one of six firms that raised their price targets on CytomX stock following the positive data for Varseta-M. The other firms included:
- Wedbush Securities (Robert Driscoll)—Up 83% from $6 to $11, maintaining “Outperform” rating
- C. Wainwright (Mitchell Kapoor)—Up 70% from $10 to $17, maintaining “Buy” rating
- Barclays (Etzer Darout)—Up 60% from $10 to $16, maintaining “Overweight” rating
- Guggenheim Securities (Michael Schmidt)—Up 50% from $10 to $15, maintaining “Buy” rating
Anupam Rama, a managing director with J.P. Morgan focused on U.S. small- and mid-capitalization biotechnology equity research, not only raised the firm’s price target 71% from $7 to $12, but also upgraded its rating on CytomX stock from “Neutral” to “Overweight.”
Lilly stock rallies after HSBC downgrade
Eli Lilly (NYSE: LLY) shares went on a rollercoaster ride Wednesday after they were downgraded from “Hold” to “Reduce” (the equivalent of “Sell”) by analysts from HSBC (London: HSBA; Hong Kong: 5), who also cut their 12-month price target on the pharma giant’s stock nearly 21%, from $1,070 to $850.
A team of analysts led by Rajesh Kumar, a managing director and head of HSBC’s European life sciences and healthcare equity research, cited three reasons for issuing what to date is Lilly’s only sell-equivalent rating for its stock.
One is their skepticism about the projected size of the total addressable market (TAM) for obesity drugs—from the blockbusters Lilly is selling now to the new treatments the company plans to bring to market starting this year, such as orforglipron. The oral small molecule glucagon-like peptide-1 (GLP-1) receptor agonist is under FDA review with a Prescription Drug User Fee Act (PDUFA) target action date of April 10.
The HSBC analysts projected the TAM to rise by 2032 to only between $80 billion and $120 billion, 20% to 53% below the $150-plus billion estimated by a consensus of other analysts. HSBC based its pessimism on the looming specter of lower prices for obesity drugs resulting from both competition, especially with archrival Novo Nordisk (NASDAQ Copenhagen: NOVO-B), direct-to-consumer sales, and Trump administration efforts such as “most favored nation” (MFN) pricing.
“The price cuts in 2026 are a headwind. The company’s guidance implies that it can continue to defy gravity with volume growth,” Kumar and colleagues wrote.
Appearing on Canada’s BNN Bloomberg TV, Kumar acknowledged Lilly’s meteoric sales growth wrought by tirzepatide, the dual-action GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) drug it markets in diabetes as Mounjaro® and in obesity as Zepbound®. Lilly finished 2025 with a near-doubling (95%) of net income from a year earlier, to $6.636 billion from $4.410 billion, on revenue that jumped 45% year-over-year, to $65.179 billion from $45.043 billion.
“Overexcited” market
“If you think about the company, their execution has been fantastic, and growth has been, I think, absolutely stunning,” Kumar said. “The problem you’ve got is, the market has gotten overexcited about the oral drug launch.”
Overexcited, Kumar argued, because oral drugs have generally shown a lower rate of medication adherence compared to injectables, as concluded by studies published in 2017 and in 2021, as well as a 2016 study in colorectal cancer patients that observed: “There should be an increased focus on improving adherence rates in patients receiving oral capecitabine.”
As a result, Kumar and his HSBC colleagues view 2026 sales forecasts for orfoglipron as too optimistic at between $1.1 billion and $1.3 billion, a range they say is anchored to the $1.5 billion in pre-launch inventories” that were “primarily related to orforglipron” as of December 31, 2025, according to the company’s Form 10-K annual report for 2025, filed February 12.
“Whilst the momentum in the launch might be positive, we think oral drug launch expectations for Lilly are too high,” the analysts wrote in their report.
But data from IMS showed a clear consumer preference for oral drugs. Oral Wegovy® (semaglutide), Novo Nordisk’s GLP-1 obesity drug, racked up 89,300 total prescriptions in its tenth week following launch on January 5, compared with 9,700 prescriptions written for injectable Wegovy, Jefferies equity analyst Akash Tewari reported in a research note.
Low-end forecast
Last year, Kumar and HSBC projected that orforglipron will generate close to $10 billion in peak-year sales, placing them at the low end of analysts’ sales forecasts.
While a consensus of analysts assembled through the Visible Alpha equities research platform has also offered a $10 billion peak-year sales forecast, David Risinger of Leerink Partners and three colleagues projected sales of $13.5 billion by 2030, while Jefferies’ Tewari has forecasted sales as high as $25 billion.
The third reason Kumar and colleagues gave for the downgrade is Lilly’s success in direct sales of some drugs through cash-based self-pay options—a success they said could be cyclical and thus short-lived.
“We think that the growth is largely driven by price rather than product differentiation. Rising working capital intensity at Lilly, headline price pressures, and rebate dynamics at both companies indicate to us that the pricing dynamics are likely to get worse,” the HSBC analysts commented.
Lilly shares dropped 7% from $930.35 to $905.11 early in Wednesday trading (9:58 a.m. ET) on news of HSBC’s downgrade. The decline was unusual for Lilly, which, like other top-tier pharmas, typically measures its day-by-day stock fluctuations in the low single digits.
But by the end of Wednesday trading, investors rallied behind Lilly enough to limit the damage to a 1.3% decline, closing at $918.05. Shares dipped a fraction (0.001%) on Thursday, closing at $917.50, and fell another 1% Friday, finishing the week at $906.70.
The downgrade marked the second time in a year that HSBC analysts lowered their rating on Lilly shares. Lilly was downgraded to “Reduce” in April 2025, with HSBC citing what it called an unattractive risk-reward profile based on what it called “too much optimism” in the company’s share price.
But by August, HSBC restored its rating to “Hold,” citing greater clarity on the commercial potential of orforglipron after Lilly released positive Phase III data for the drug.
Leaders and laggards
- Cingulate (NASDAQ: CING) shares nosedived 31% from $11.66 to $8.04 Wednesday after the developer of therapies based on its PTR drug delivery platform reported a larger-than-expected fourth quarter net loss of $6.273 million, vs. a $6.231 million net loss in Q4 2024. Cingulate finished 2025 with a net loss of $22.45 million, up from a net loss of $16.56 million a year earlier. A consensus of analysts projected a $4.8 million loss for Q4 and a $20.8 million loss for all of last year. Cingulate blamed its increased losses on higher general and administrative expenses plus interest on notes payable. The company cautioned that its cash and cash equivalents shrunk 11% to $11 million as of December 31, 2025, from $12.3 million a year earlier, only enough cash to satisfy current capital needs into the late fourth quarter. Despite the setback, Cingulate shares have soared 80% year-over-year, from $3.64 on March 18, 2025.
- Ovid Therapeutics (NASDAQ: OVID) shares climbed 34% over four trading days, immediately before and after reporting positive topline Phase I safety, tolerability, and pharmacokinetics findings from the 7 mg dose cohort of OV329, its next-generation GABA-aminotransferase (GABA-AT) inhibitor being developed to treat drug-resistant epilepsies. As a result, Ovid plans to advance OV329 into a Phase II trial in focal onset seizures and an open-label, proof-of-concept study. Ovid said it will add complementary development programs for OV329, expanding into tuberous sclerosis complex seizures and infantile spasms—an expansion to be funded through a private placement financing expected to generate $60 million in gross proceeds before deducting placement agent fees and offering expenses. Shares jumped from $1.95 Monday to $2.62 at the close of trading Friday, fueled by surges of 14% Wednesday (from $2.01 to $2.30) and 14% Friday (from $2.30 to $2.62).
The post StockWatch: CytomX Shares Yo-Yo on Colorectal Cancer Data appeared first on GEN – Genetic Engineering and Biotechnology News.
GEN – Genetic Engineering & Biotechnology News
Bioengineered Implants Deliver Multi-Drug Therapy in Animal Models
In a new paper, scientists from Northwestern University and their collaborators at Rice University and Carnegie Mellon University report on their progress towards developing so-called implantable “living pharmacies.” These are tiny devices containing engineered cells that continuously produce medicines inside the body. Details of the study, which was done in rats, are published in Device in a paper titled “Design of a wireless, fully implantable platform for in-situ oxygenation of encapsulated cell therapies.”
The device, which is called the hybrid oxygenation bioelectronics system for implanted therapy or HOBIT, is roughly the size of a folded stick of gum. It integrates engineered cells with oxygen-producing bioelectronics and is designed in such a way that the cells are shielded from the body’s immune system while also receiving oxygen and nutrients needed to keep them alive and producing drugs for several weeks. In the future, these devices could be deployed to treat chronic conditions without requiring patients to carry, inject, or remember to take medications.
“This work highlights the broad potential of a fully integrated biohybrid platform for treating disease,” said Jonathan Rivnay, PhD, a professor of biomedical engineering and materials science and engineering at Northwestern and a co-principal investigator of the project. “Traditional biologic drugs often have very different half-lives, so maintaining stable levels of multiple therapies can be challenging. Because our implanted ‘cell factories’ continuously produce these biologics, keeping the cells alive with our oxygenation technology allows us to sustain steady levels [of] multiple different therapeutics at once.”
Solving the oxygenation challenge was critical to the success of HOBIT. When engineered cells are packed together in an implant, they compete for oxygen to live. Without a steady supply, many cells die, which limits how much medicine the implants can produce. In an earlier study, Rivnay and his collaborators demonstrated how a tiny electrochemical device could generate oxygen by splitting nearby water molecules, and showed that supplying oxygen locally dramatically improved the survival of implanted therapeutic cells. The latest iteration of their device integrates that oxygen-generation technology in a fully implantable, wireless system.
Digging into the details of the device, HOBIT contains three primary components: a cell chamber that holds the genetically engineered cells, a miniature oxygen generator, and electronics and a battery to regulate oxygen production and wirelessly communicate with external devices. Because the device produces oxygen directly inside the implant, the cells receive a steady supply even in hypoxic environments. “We are producing oxygen directly where the cells need it,” Rivnay said. “That allows us to support much higher cell densities in a much smaller space.” In fact, “cell densities in HOBIT were roughly six times higher than conventional unoxygenated encapsulation approaches.”
According to the paper, the team engineered the cells to produce three different biologics—an anti-HIV antibody, a GLP-1-like peptide used to treat type 2 diabetes, and leptin, a hormone that regulates appetite and metabolism. They implanted the devices under the skin of rats and monitored drug levels in their bloodstreams for 30 days. Blood measurements of animals with the implanted devices showed sustained levels of all three biologics throughout the study period. In contrast, in animals that were implanted with devices without oxygenation, the biologics that had shorter half-lives were undetectable by the seventh day. Drugs with longer half-lives in these animals also declined steadily over time. At the end of the testing period, roughly 65% of the cells in the oxygenated devices remained viable compared with roughly 20% in control devices.
For their next steps, the scientists intend to test their devices in larger animal models and explore disease-specific applications, including therapies based on transplanted pancreatic cells. “As these technologies continue to develop, devices like this could eventually act as programmable drug factories inside the body—delivering complex therapies in ways that simply aren’t possible today,” Rivnay said.
The post Bioengineered Implants Deliver Multi-Drug Therapy in Animal Models appeared first on GEN – Genetic Engineering and Biotechnology News.
GEN – Genetic Engineering & Biotechnology News
Gut-Immune Link Identified in Multiple Sclerosis-Related Neuroinflammation
Multiple sclerosis (MS) is a debilitating neurological disorder caused by malfunctioning immune responses that target the brain and spinal cord of the central nervous system (CNS). New research led by Shohei Suzuki, MD, PhD, assistant professor, division of gastroenterology and hepatology, and Tomohisa Sujino, PhD, associate professor, School of Medicine, at Keio University, Japan, has now indicated how the gut can initiate neuroinflammation in multiple sclerosis.
Their study found that intestinal epithelial cells (IECs) promote the development of pathogenic T cells that migrated to the spinal cord and induced disease symptoms in mouse models of the disorder.
The researchers examined intestinal tissues from patients with MS and mice with experimental autoimmune encephalomyelitis (EAE), a close analog of MS. In both cases, they observed an increase in TH17 cells and an upregulation of major histocompatibility complex class II (MHC II) expression in IECs. Deleting MHC II in IECs reduced the accumulation of TH17 cells in the gut and lowered the severity of EAE. They suggest the results could inform future strategies for developing targeted therapeutics against autoimmunity.
“While current therapies for MS often target B cells, our study highlights the gut as an important therapeutic site,” Suzuki commented. “Modulating intestinal microbiota or antigen-presenting activity of IECs represents new approaches to treating autoimmune neurological diseases.”
Suzuki, Sujino, and colleagues reported on their findings in Science Immunology, in a paper titled “Intestinal Epithelial MHC Class II Induces Encephalitogenic CD4⁺ T Cells and Initiates Central Nerves System Autoimmunity,” in which they concluded, “Our findings reveal an interaction between gut IECs and neuroinflammatory diseases through MHC II expression in human MS and mouse EAE, providing a mechanistic link between gut immune education and CNS autoimmunity and opening new avenues for targeting intestinal immunity in neuroinflammatory diseases.
Failure of the immune system to distinguish ‘self’ from ‘non-self’ entities leads to excessive autoimmune responses against self-proteins like myelin, which forms a protective covering on the neurons. Multiple factors influence the onset and progression of MS, including genetic susceptibility, environmental triggers, and, more recently, the gut microenvironment. Patients with MS exhibit alterations in their gut microbiota, while the gut microbiota and microbial metabolites play a pivotal role in shaping the chronic autoreactive immune responses. “… in an experimental autoimmune encephalomyelitis (EAE) model, commensal or specific microbes were found to be essential for disease initiation and progression,” the authors wrote.
However, in trying to define this gut–CNS axis, the cellular mechanisms that relay the gut-derived signals to the immune system to influence autoimmune inflammation in the CNS remain poorly understood. “Increasing evidence shows that the gut microbiota influences neurological diseases such as Parkinson’s, Alzheimer’s, and MS,” Sujino stated. “However, the mechanisms linking gut microbes, intestinal immunity, and brain inflammation remain unclear. We were keen to identify how gut immune responses contribute to neuroinflammatory diseases.”
Prior research has shown that gut-derived signals can promote the differentiation of T cells into pathogenic T helper 17 (TH17) in mouse models of MS. Recent studies have suggested that IECs can function as antigen presenting cells that help induce these pathogenic cells, but the underlying mechanisms have been unclear.
Building on their previous observation that mild intestinal (ileal) inflammation exists in experimental autoimmune encephalomyelitis (EAE), which is a mouse model of MS, the authors set out to test whether similar inflammation is present in patients with MS. By performing single-cell RNA sequencing on intestinal biopsies, the team identified that inflammatory Th17 cells accumulate in the mouse EAE model as well as in the intestine of patients with MS, suggesting a conserved gut–CNS axis that may be active in human diseases.
In both EAE mice and patients with MS, intestinal epithelial cells upregulated antigen presentation pathways. Particularly, epithelial cells in the ileum had higher expression of major histocompatibility complex class II (MHC II) that presents antigens to CD4+ T cells. “Clinically, patients with MS exhibited an increased expression of epithelial MHC II–associated genes and an accumulation of CD4 T cells in the small intestine, suggesting the conservation of this gut-CNS axis in human diseases,” the scientists stated. Experiments showed that selective deletion of MHC II in IECs reduced pathogenic Th17 cell generation and disease severity. “Conditional deletion of MHC II in IECs showed that epithelial antigen presentation was indispensable for the local expansion of pathogenic Th17 cells in the gut and their subsequent migration to the CNS,” the team stated.
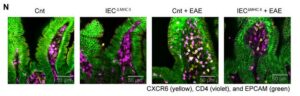
![Immunofluorescence analysis was performed on terminal ileum samples from Cnt, IECΔMHCII, Cnt + EAE, and IECΔMHCII + EAE mice. A total of 3–5 tissue sections were analyzed per mouse, with 3 mice included in each group. [Shohei Suzuki]](https://www.genengnews.com/wp-content/uploads/2026/03/low-res-2-1-300x96.jpeg)
mouse, with three mice included in each group. [Shohei Suzuki]
IECs do not typically present antigens to immune cells. So, the team conducted co-culture assays to test the antigen presentation function of IECs. Their findings demonstrate that IECs can directly present antigens in an MHC II-dependent manner to prime CD4+ T cells in the gut. Notably, in these assays, IECs induced Th17 polarization of activated CD4+ T cells. It became clear that the gut was a critical site for immune activation of pathogenic CD4+ T cells that polarized into pro-inflammatory Th17 cells. “These findings provide direct functional evidence that IEC-expressed MHC II is sufficient to drive Th17 polarization from primed CD4 T cells in an antigen-dependent manner, supporting a direct role for IECs as non-professional antigen-presenting cells,” the scientists reported.
To investigate whether the Th17 cells directly contribute to the pool of autoreactive cells in the CNS, they used transgenic mice that express the Kaede protein, which undergoes photoconversion from green to red fluorescence upon exposure to violet light. This model allowed for precise tracking of pathogenic Th17 cells induced in the intestinal lamina propria that then migrate to the spinal cord and drive neuroinflammation.
Taken together, the study findings reveal a critical role for MHC II expressed by IECs in the expansion of pathogenic Th17 cells that subsequently migrate to the CNS during EAE, providing a mechanistic link between gut immune responses and autoimmune neuroinflammatory diseases. The results demonstrate that while systemic circulation allows T cell exchange across immune tissues, the epithelial–immune interactions within the gut mucosal compartment can essentially shape effector T cell responses in the brain.
“This study reveals a previously unknown role of IECs in antigen presentation and Th17 programming, thereby defining a gut-CNS immunological axis with important implications for understanding and treating autoimmune neuroinflammation,” the authors concluded. “Our findings suggest that the modulation of epithelial antigen presentation could serve as a novel therapeutic approach for MS and related diseases. Given the accessibility of the gut epithelium to dietary, microbial, and pharmacological interventions, targeting IEC–T cell interactions may offer a tractable strategy for immunomodulation.”
The post Gut-Immune Link Identified in Multiple Sclerosis-Related Neuroinflammation appeared first on GEN – Genetic Engineering and Biotechnology News.
GEN – Genetic Engineering & Biotechnology News
Agentic AI, Virtual Cell, LNP Vaccine Boosters, Engineered Organs, and Mergers
This week, agentic AI steps into the limelight buoyed by the momentum from generative AI. And there’s a new virtual cell model in town courtesy of AI-drug developer Xaira Therapeutics. From the frontiers of AI, our discussion turned to feats of engineering in regenerative medicine and lipid nanoparticles. In one study, scientists redesigned LNPs to avoid the liver and accumulate in the lymph nodes. In the other, efforts to develop and implant a lab grown esophagus from donor pigs bear fruit. Finally, Novartis plans to spend up to $3 billion to expand its cancer pipeline with the acquisition of Pikavation Therapeutics. And Merck is acquiring Terns Pharmaceuticals for approximately $6.7 billion also with an eye towards boosting its cancer portfolio.
Listed below are links to the GEN stories referenced in this episode of Touching Base:
NVIDIA GTC 2026: Agentic AI Inflection Hits Healthcare and Life Sciences
By Fay Lin, PhD, GEN Edge, March 18, 2026
Xaira’s First Virtual Cell Model Is Largest To-Date, Toward Complex Biology
By Fay Lin, PhD, GEN Edge, March 25, 2026
Modified Lipid Nanoparticles Boost mRNA Vaccine Delivery to Lymph Nodes
GEN, March 24, 2026
Engineered Esophagus Rebuilds Missing Organ Segment in Pig Models
GEN, March 20, 2026
Novartis Acquires Pikavation for Up to $3B, Expanding Cancer Pipeline
GEN, March 22, 2026
Merck Bolsters Cancer Pipeline with $6.7B Terns Buyout
By Alex Philippidis, GEN Edge, March 25, 2026
Touching Base Podcast
Hosted by Corinna Singleman, PhD
Behind the Breakthroughs
Hosted by Jonathan D. Grinstein, PhD
The post Agentic AI, Virtual Cell, LNP Vaccine Boosters, Engineered Organs, and Mergers appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors




