Uncategorized
New protein drugs could pave way for faster drug development
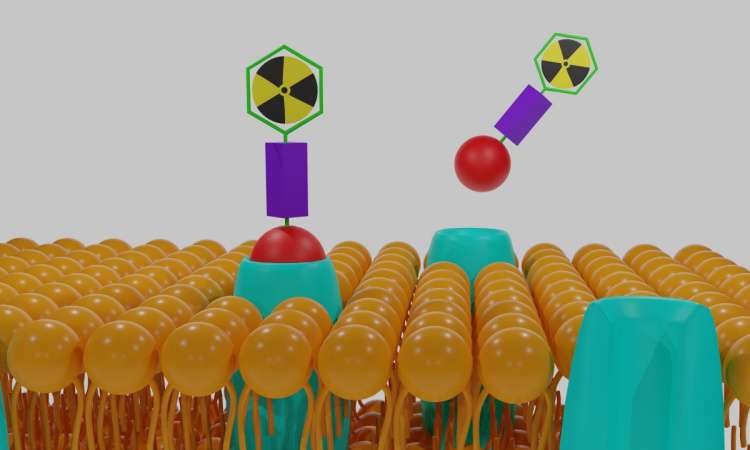

A new class of protein drugs have shown amenability to multiple isotopes, paving the way for faster radiopharmaceutical drug development.
Biotechnology company Molecular Partners announced preclinical data showing its Radio-DARPins are suitable to different isotopes, each specific for a different tumour target.
DARPin (Designed Ankyrin Repeat Protein) therapeutics are a novel class of protein drugs based on natural binding proteins, which have been clinically validated across several therapeutic areas and developed through to the registrational stage.
The key properties of DARPins – intrinsic high affinity and specificity, small size, flexible architecture, and high stability – offer unmatched advantages to drug design, such as multi-specificity, broad target range, and tuneable half-life.
The results of studies in tumour-bearing mice show highly comparable biodistribution profiles for both Radio-DARPin candidates labelled with Lutetium-177 (177Lu) or with Lead-203 (203Pb), with similar uptake and washout rates. Imaging with 177Lu can be indicative of behaviour with the therapeutic isotope Actinium-225 (225Ac), and similarly with 203Pb for 212Pb.
The preclinical data was revealed at the Global Radiopharmaceuticals Development Summit, which took place in Shanghai, China on March 19–20.
“Our recent data confirms that our Radio-DARPin-vector design allows interchangeability of alpha-isotopes, including 212Pb and 225Ac,” said Patrick Amstutz, CEO of Molecular Partners.
“This feature offers us the opportunity and flexibility to evaluate Radio-DARPin candidates in an isotope-agnostic manner and to choose the most suitable therapeutic isotope, as late as with initial clinical data, without having to restart the entire drug discovery and development process – a significant advantage to tailor our candidates to patient needs.”
The post New protein drugs could pave way for faster drug development appeared first on Drug Discovery World (DDW).
Uncategorized
High-Throughput Platform for Fast-Acting Covalent Protein Therapies
Researchers at Westlake University in China, lead by Bobo Dang, PhD, and Ting Zhou, PhD, report the development of a high-throughput platform for engineering fast-acting covalent protein therapeutics. The team says their study “A high-throughput selection system for fast-acting covalent protein drugs,” published in Science, opens new avenues for next-generation biologics.
Covalent small-molecule drugs have shown great success in cancer therapy by forming irreversible bonds with their targets. This has inspired efforts to extend covalent strategies to protein therapeutics, especially engineered miniproteins. However, their development is limited by a kinetic mismatch. Miniproteins are rapidly cleared in vivo, while covalent bond formation is typically slow. In addition, high-throughput platforms for systematically optimizing covalent protein reactivity have been lacking.
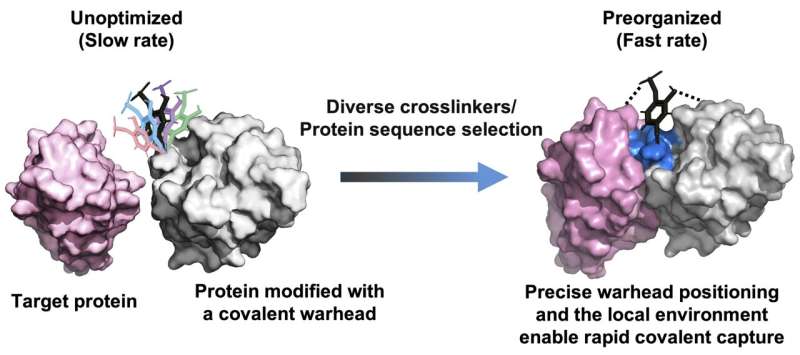
To address this challenge, the researchers proposed that precise spatial positioning of chemical warheads within protein scaffolds could enable molecular preorganization, thereby accelerating covalent bond formation without increasing intrinsic reactivity (see figure).
![The principle for developing fast-acting covalent proteins via comprehensive crosslinker and protein sequence engineering. [Bobo Dang's Lab at Westlake University]](https://www.genengnews.com/wp-content/uploads/2026/04/fast-acting-covalent-p.jpg)
Based on this concept, the team created a high-throughput platform that combines yeast surface display with chemoselective protein modification to screen diverse crosslinkers and millions of protein variants. The platform enables rapid and irreversible target engagement.
Using this platform, the researchers developed a covalent antagonist targeting PD-L1, termed IB101. Structural analysis revealed that IB101 forms a defined binding pocket that precisely positions the active moiety in a reactive conformation, greatly accelerating covalent bond formation.
Functionally, IB101 effectively blocks the PD-1/PD-L1 immune checkpoint pathway and demonstrates strong antitumor activity in mouse models. Notably, despite its short in vivo half-life, IB101 achieves durable target engagement and tumor suppression, outperforming conventional antibody-based therapies under comparable conditions, according to the scientists.
The platform was further applied to cytokine engineering, leading to the development of a covalent IL-18 variant, IB201. This engineered cytokine rapidly forms a covalent interaction with its receptor, enhancing signaling strength and duration. In vivo studies showed that IB201 induces potent antitumor immune responses without detectable systemic toxicity. These results highlight the potential of covalent engineering to improve the efficacy and safety of cytokine-based therapies.
Beyond immunotherapy targets, the platform was also applied to develop a covalent inhibitor targeting the receptor-binding domain (RBD) of SARS-CoV-2. This molecule showed durable viral neutralization, demonstrating the versatility of the approach across different therapeutic modalities, note the researchers, adding that the study establishes a general strategy for engineering fast-acting covalent protein therapeutics.
By enabling covalent bond formation on timescales compatible with rapid in vivo clearance, the platform overcomes a fundamental limitation in the field, say the scientists. These findings, they continue, provide a new framework for designing biologics with both rapid kinetics and sustained target engagement, with broad implications for cancer immunotherapy, antiviral therapy, and beyond.
The post High-Throughput Platform for Fast-Acting Covalent Protein Therapies appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Terns rebuffed a higher bid before selling to Merck

Regulatory filings show that Terns ultimately accepted an offer 15% lower than a previous proposal as four companies vied for rights to the coveted leukemia drug developer.


Regulatory filings show that Terns ultimately accepted an offer 15% lower than a previous proposal as four companies vied for rights to the coveted leukemia drug developer.
Uncategorized
STAT+: States looking to regulate use of chatbots
You’re reading the web edition of STAT’s Health Tech newsletter, our guide to how technology is transforming the life sciences. Sign up to get it delivered in your inbox every Tuesday and Thursday.
Good morning health tech readers!
Today, a deep dive into why America’s most powerful health insurer is looking more and more like a technology company.

You’re reading the web edition of STAT’s Health Tech newsletter, our guide to how technology is transforming the life sciences. Sign up to get it delivered in your inbox every Tuesday and Thursday.
Good morning health tech readers!
Today, a deep dive into why America’s most powerful health insurer is looking more and more like a technology company.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors