Uncategorized
Microplastics in Human Bile Drive Mitochondrial Dysfunction and Senescence
Microplastics have become a defining environmental signature of modern life, turning up in oceans, soil, food, drinking water, and even the air. But their biological fate inside the human body remains far less understood. A new study suggests that these particles may be doing more than simply passing through. Instead, they may be accumulating in one of the body’s most overlooked fluids—bile—and leaving behind measurable cellular damage that could shape future thinking about environmentally driven biliary injury and long‑term health effects. As the authors noted in their abstract, “the long-term accumulation patterns and chronic toxic effects of microplastics within the human biliary system are largely unknown,” underscoring the need for deeper investigation into how these particles behave in the enterohepatic circulation.
Researchers from the Tenth Affiliated Hospital of Southern Medical University (Dongguan People’s Hospital), Sun Yat-sen University, Guilin Medical University, and collaborating institutions reported the findings in Environmental Science and Ecotechnology. Their study, “Microplastics accumulate in human bile and drive cholangiocyte senescence,” provides the first direct evidence that microplastics are not only present in bile but may also contribute to mitochondrial dysfunction and premature aging in cholangiocytes, the epithelial cells that line the bile ducts.
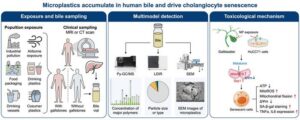
The team collected bile from 14 surgical patients (five without gallstones and nine with gallstones) and used a multimodal analytical approach—pyrolysis–gas chromatography–mass spectrometry, laser direct infrared spectroscopy, and scanning electron microscopy—to characterize the particles. According to the paper, “we show the universal presence of microplastics in human bile,” identifying six polymer types dominated by polyethylene terephthalate and polyethylene, with most particles measuring 20–50 μm. Patients with gallstones carried substantially higher microplastic burdens, raising questions about whether biliary stasis or altered bile composition may influence microplastic retention.

To probe biological effects, the researchers exposed cultured human cholangiocytes to low-dose polystyrene nanoplastics for seven days, simulating chronic exposure. The cells exhibited mitochondrial dysfunction, elevated reactive oxygen species, reduced ATP, Drp1‑mediated mitochondrial fission, and G1 cell‑cycle arrest—hallmarks of senescence. As the authors wrote, chronic exposure “induces mitochondrial dysfunction-associated senescence in cholangiocytes,” suggesting a mechanistic link between environmental microplastics and biliary aging.
One of the most intriguing findings is that melatonin, a widely used antioxidant, partially reversed the mitochondrial and inflammatory damage. While far from a therapeutic recommendation, the result hints at a potential intervention point and gives the study translational relevance.
The work reframes the biliary system as something far more active than a simple transit channel. The data indicate that bile can serve as a reservoir for microplastics and that prolonged exposure may age cholangiocytes by driving mitochondrial dysfunction. The partial rescue with melatonin adds a mechanistic foothold for future intervention, even as the authors caution that broader human studies are essential.
For biotech, the implications are broad. The work highlights bile as a clinically accessible matrix for exposure assessment, opening the door to new diagnostics for environmental toxicology. The mitochondrial stress signature aligns with pathways already being targeted by companies developing senolytics, mitoprotective agents, and anti‑inflammatory therapeutics. The authors wrote that the research provides “a mechanistic foundation for assessing the health risks of plastic pollution and developing therapeutic interventions for environmentally driven biliary disorders.”
The post Microplastics in Human Bile Drive Mitochondrial Dysfunction and Senescence appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
STAT+: Health insurers score major win with higher 2027 Medicare Advantage rates
Companies that sell Medicare Advantage plans will receive a 2.5% pay bump on average in 2027, up significantly from what was proposed and a win for an industry that has experienced higher medical costs and has opposed nearly all reforms to the lucrative taxpayer-financed program.
More importantly, the Trump administration scrapped its proposal that would have used more updated data in the payment process, ensuring that Medicare Advantage insurers retain billions of dollars.
In addition to base payments, Medicare Advantage insurers get paid based on how sick their members. This process is known as risk adjustment and has been flagged by watchdogs, auditors, and federal attorneys as rife with abuse. Trump officials proposed using newer data that goes into seniors’ “risk scores,” but after intense industry pushback over the past two months, they are dropping the proposal for now.

Companies that sell Medicare Advantage plans will receive a 2.5% pay bump on average in 2027, up significantly from what was proposed and a win for an industry that has experienced higher medical costs and has opposed nearly all reforms to the lucrative taxpayer-financed program.
More importantly, the Trump administration scrapped its proposal that would have used more updated data in the payment process, ensuring that Medicare Advantage insurers retain billions of dollars.
In addition to base payments, Medicare Advantage insurers get paid based on how sick their members. This process is known as risk adjustment and has been flagged by watchdogs, auditors, and federal attorneys as rife with abuse. Trump officials proposed using newer data that goes into seniors’ “risk scores,” but after intense industry pushback over the past two months, they are dropping the proposal for now.
Uncategorized
CRISPR at 25: The Past, Present, and Future of Genome Editing

Panelists:

Rodolphe Barrangou, PhD
North Carolina State University
Panelist

Rodolphe Barrangou, PhD
Rodolphe Barrangou, PhD, is the T. R. Klaenhammer Distinguished Professor at North Carolina State University, where he leads the CRISPR Lab. Rodolphe spent nine years at Danisco and DuPont, where he made seminal contributions in the functional characterization of CRISPR as a microbial immune system. He has been at NC State since 2013.
For his CRISPR work, Rodolphe has received several international awards, notably the Canada Gairdner International Award, and has been elected to the National Academy of Sciences, the National Academy of Engineering, and the National Academy of Inventors. Rodolphe is a scientific co-founder of Intellia Therapeutics, Locus Biosciences, TreeCo, Ancilia Biosciences, and CRISPR Biotechnologies, and an advisor to Inari and the IGI. He is also the founding Editor in Chief of The CRISPR Journal (published by Mary Ann Liebert, Inc., a Sage partner), which launched in 2018.
Rodolphe holds a degree from Paris Descartes University and a PhD in functional genomics from NC State.
- Time:
It has been almost 25 years since the acronym “CRISPR” was first coined. Since then, CRISPR has become a household word, a star of books and films, and a Nobel Prize–winning discovery. This powerful and disruptive genome editing technology has transformed countless fields, including gene therapy, xenotransplantation, de-extinction and agbiotech. Researchers continue to build on the CRISPR chassis, devising new platforms for bespoke genome editing. But major questions remain around clinical safety, commercial development, ethical deployment, and regulatory oversight.
In the first of a new series of GEN Keynote Webinars, Professor Rodolphe Barrangou, PhD (North Carolina State; EIC, The CRISPR Journal) offers a front-row perspective of the CRISPR revolution, the seminal advances, clinical highlights, and rising applications. Almost two decades ago, Barrangou provided the first experimental demonstration of the functional role of CRISPR. With numerous advisory and entrepreneurial activities in the gene editing space, Barrangou is the ideal guide to discuss CRISPR’s progress in the clinic; the state of the CRISPR toolbox; and the regulatory roadblocks and ethical challenges that will shape the application of CRISPR in agbiotech, germline editing, and other arenas.
Registration for this GEN Keynote Webinar is free. Following this live presentation, Dr. Barrangou will answer audience questions.
Produced with support from:
The post CRISPR at 25: The Past, Present, and Future of Genome Editing appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Immunotherapy Enhanced by Restoring Mitochondrial Function in Dendritic Cells
In a new study published in Science titled, “Mitochondrial metabolism and signaling direct dendritic cell function in antitumor immunity,” researchers from St. Jude Children’s Research Hospital have uncovered a new metabolic mechanism for how tumors disable immune “gatekeeper” cells that initiate response in the presence of cancer. The results offer a new path to improve immunotherapy.
Dendritic cells alert and activate tumor-killing immune cells as a critical part of anticancer immune response. The authors found that tumors reduce dendritic cell function by minimizing mitochondrial fitness to prevent anticancer immune response. Correspondingly, boosting mitochondrial function in dendritic cells enhances antitumor immune activity and strengthens the efficacy of existing immunotherapies.
Within the nutrient-sparse tumor microenvironment, dendritic cells progressively lose mitochondrial activity, which drives cell dysfunction and weakens immune defenses against cancer. When dendritic cells with high mitochondrial activity were introduced into tumors in preclinical mouse models, results showed that immunogenic activity was restored while improving tumor control.
“We found that tumors reprogram mitochondrial metabolism in dendritic cells, reducing their ability to activate the immune system against cancer,” said Hongbo Chi, PhD, St. Jude Department of Immunology chair and corresponding author of the study. “By enhancing mitochondrial function, we could restore dendritic cell activity and rescue antitumor immunity.”
Immunotherapies for cancer, such as immune checkpoint blockade, have greatly improved care for many malignancies, but have not been successful in all cancers. To determine whether these findings could improve immunotherapy effectiveness in tumor-bearing mice, the authors evaluated the administered dendritic cells with high mitochondrial activity in combination with immune checkpoint blockade.
“We saw the most pronounced therapeutic effect in mice treated with the combination of dendritic cells that had high mitochondrial activity and immune checkpoint blockade,” said co-first author Zhiyuan You, PhD, researcher at St. Jude Department of Immunology. “Those combinations synergistically slowed or stopped tumor growth and extended survival far more than either treatment alone.”
To test long-term effects, the researchers exposed combination therapy treated mice to a new tumor after a few months. New tumor growth stopped for these mice, indicating durable, long-term immune memory.
To better understand the relationship between mitochondrial function and dendritic cells, the researchers examined metabolic pathways affected by the tumor microenvironment. They identified a signaling axis composed of two proteins, OPA1 and NRF1, that regulate communication between mitochondria and the nucleus. Expression was greatly downregulated in dendritic cells during tumor progression and acted as a metabolic switch to shut down dendritic cell immunogenic activity.
“We’re seeing a direct regulation of dendritic cells by the tumor microenvironment,” said co-first author Jiyeon Kim, PhD, researcher at St. Jude Department of Immunology. “We have characterized how that results in mitochondrial reprogramming of dendritic cells to benefit cancer, giving us new opportunities to reverse the process.”
The study’s mechanistic insights enable new directions to rewire dendritic cell function and enhance cancer treatments.
“These findings reinforce the central role of dendritic cells in cancer immunity,” Chi said. “By exploring their mitochondrial function in the tumor microenvironment, we have provided a proof-of-principle of how we may be able to improve the next generation of immunotherapies.”
The post Immunotherapy Enhanced by Restoring Mitochondrial Function in Dendritic Cells appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors