Uncategorized
Epigenetic Strategy Restores Tumor Suppressor in Acute Myeloid Leukemia Models
Scientists from The Jackson Laboratory (JAX) and their collaborators elsewhere have found a potential way to treat cases of acute myeloid leukemia that involves turning a key cancer fighting gene back on. Besides potentially treating AML without harsh chemotherapy regimens, their work also highlights a promising strategy for studying gene-silencing mechanisms in other diseases. Full details of the study, which was done in mice, are available in a paper published in Science Translational Medicine titled “Epigenetic reactivation of the tumor suppressor ZBTB7A by KDM4 inhibition in human acute myeloid leukemia.”
Normally, tumor suppressor genes work to prevent cells from becoming cancerous. But in cancers like AML, some of these genes are switched off epigenetically. These changes to gene activity are difficult to track because standard DNA sequencing technologies are designed to find mutated DNA. “If we can identify which genes have been silenced and understand how to turn them back on, that could open up entirely new therapeutic possibilities,” said Eric Wang, PhD, an assistant professor JAX who led the research. “Instead of only trying to kill these cells, we may be able to restore the mechanisms that normally keep them under control.”
Though scientists have made great strides in developing therapies for AML, prognosis for the disease is still relatively poor. Part of the challenge is that AML cells remain in an immature, stem cell-like state. According to the paper, Wang and his team developed a tool that combines fluorescence in situ hybridization and flow cytometry with CRISPR gene editing technology to map gene activity in cells. They used the tool, called FISHnCRISP, to identify a tumor-suppressing gene called ZBTB7A that is silenced in AML patients. By restoring ZBTB7A expression, the scientists forced the cancer cells into a state where they grew less aggressively.
Digging into the details, AML cells produce a longer version of ZBTB7A’s regulatory tail, that contains sites that attract a protein called ZFP36L2, which reduces the gene’s activity. Additionally, a family of enzymes known as KDM4 modify how DNA is packaged inside AML cells, which effectively silences ZBTB7A expression. Data from experiments in mice with AML showed that when KDM4 enzymes were blocked, ZBTB7A regained its expression, reducing leukemia burden while leaving normal blood formation largely unaffected.
Importantly, “there are drug candidates out there to inhibit KDM4, and in our study we just repurposed one of them to treat AML cells,” Wang said. “We won’t know unless we test it in clinical trials, but this approach could be better than chemotherapy, because we showed it’s not toxic at all to normal blood cells.”
Future studies will focus on refining the approach and determining whether it might be combined with existing treatments. The team plans to test an experimental drug that targets KDM4, which is currently being tested in a clinical trial for solid tumors.
“We demonstrated that downregulating ZBTB7A causes this hyperinflammatory state that promotes cancer growth” and “now, we’re proposing this epigenetic approach to force AML cells to differentiate into white blood cells that eventually undergo cell death,” Wang said. “We could potentially translate our research into an early phase clinical trial more readily than developing a whole new compound from scratch.”
The post Epigenetic Strategy Restores Tumor Suppressor in Acute Myeloid Leukemia Models appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Opinion: STAT+: Former Geisinger CEO: U.S. health systems must replace huge numbers of people with AI
About 20 years ago, I stepped on stage at one of our Geisinger town halls and looked out upon a sea of people: thousands of full-time employees at an integrated health system charged with the health and well-being of millions of Pennsylvanians.
Only a fraction of the people in that room were clinicians.
That was the first time I fully visualized the problem: We employed more people in our revenue cycle department to process bills and reconcile data than we did doctors. And we weren’t alone. It’s the same story at every health system in America, large and small, and over the past two decades, the ratio has become dramatically more disparate.

About 20 years ago, I stepped on stage at one of our Geisinger town halls and looked out upon a sea of people: thousands of full-time employees at an integrated health system charged with the health and well-being of millions of Pennsylvanians.
Only a fraction of the people in that room were clinicians.
That was the first time I fully visualized the problem: We employed more people in our revenue cycle department to process bills and reconcile data than we did doctors. And we weren’t alone. It’s the same story at every health system in America, large and small, and over the past two decades, the ratio has become dramatically more disparate.
Uncategorized
A star scientist showed that better genetics lessons could reduce racism. It was the death knell for his career
Every year, the Genetics Society of America bestows the Elizabeth W. Jones Award for Excellence in Education, recognizing someone who has helped the public better understand the science of DNA. It’s understood to be a lifetime achievement award; past recipients tend toward retirement age with decades of work behind them and stacks of textbooks to their names.
When this year’s winner, Brian Donovan, was announced at the end of February, many geneticists and science educators found it hard to celebrate the news. Not because he’s undeserving of the honor. Far from it. But because it seemed to confirm what many feared: that Donovan’s incandescent research career was over before it had barely begun.

Every year, the Genetics Society of America bestows the Elizabeth W. Jones Award for Excellence in Education, recognizing someone who has helped the public better understand the science of DNA. It’s understood to be a lifetime achievement award; past recipients tend toward retirement age with decades of work behind them and stacks of textbooks to their names.
When this year’s winner, Brian Donovan, was announced at the end of February, many geneticists and science educators found it hard to celebrate the news. Not because he’s undeserving of the honor. Far from it. But because it seemed to confirm what many feared: that Donovan’s incandescent research career was over before it had barely begun.
Uncategorized
Microplastics in Human Bile Drive Mitochondrial Dysfunction and Senescence
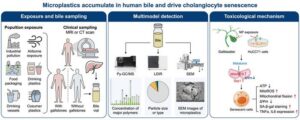
Microplastics have become a defining environmental signature of modern life, turning up in oceans, soil, food, drinking water, and even the air. But their biological fate inside the human body remains far less understood. A new study suggests that these particles may be doing more than simply passing through. Instead, they may be accumulating in one of the body’s most overlooked fluids—bile—and leaving behind measurable cellular damage that could shape future thinking about environmentally driven biliary injury and long‑term health effects. As the authors noted in their abstract, “the long-term accumulation patterns and chronic toxic effects of microplastics within the human biliary system are largely unknown,” underscoring the need for deeper investigation into how these particles behave in the enterohepatic circulation.
Researchers from the Tenth Affiliated Hospital of Southern Medical University (Dongguan People’s Hospital), Sun Yat-sen University, Guilin Medical University, and collaborating institutions reported the findings in Environmental Science and Ecotechnology. Their study, “Microplastics accumulate in human bile and drive cholangiocyte senescence,” provides the first direct evidence that microplastics are not only present in bile but may also contribute to mitochondrial dysfunction and premature aging in cholangiocytes, the epithelial cells that line the bile ducts.
The team collected bile from 14 surgical patients (five without gallstones and nine with gallstones) and used a multimodal analytical approach—pyrolysis–gas chromatography–mass spectrometry, laser direct infrared spectroscopy, and scanning electron microscopy—to characterize the particles. According to the paper, “we show the universal presence of microplastics in human bile,” identifying six polymer types dominated by polyethylene terephthalate and polyethylene, with most particles measuring 20–50 μm. Patients with gallstones carried substantially higher microplastic burdens, raising questions about whether biliary stasis or altered bile composition may influence microplastic retention.

To probe biological effects, the researchers exposed cultured human cholangiocytes to low-dose polystyrene nanoplastics for seven days, simulating chronic exposure. The cells exhibited mitochondrial dysfunction, elevated reactive oxygen species, reduced ATP, Drp1‑mediated mitochondrial fission, and G1 cell‑cycle arrest—hallmarks of senescence. As the authors wrote, chronic exposure “induces mitochondrial dysfunction-associated senescence in cholangiocytes,” suggesting a mechanistic link between environmental microplastics and biliary aging.
One of the most intriguing findings is that melatonin, a widely used antioxidant, partially reversed the mitochondrial and inflammatory damage. While far from a therapeutic recommendation, the result hints at a potential intervention point and gives the study translational relevance.
The work reframes the biliary system as something far more active than a simple transit channel. The data indicate that bile can serve as a reservoir for microplastics and that prolonged exposure may age cholangiocytes by driving mitochondrial dysfunction. The partial rescue with melatonin adds a mechanistic foothold for future intervention, even as the authors caution that broader human studies are essential.
For biotech, the implications are broad. The work highlights bile as a clinically accessible matrix for exposure assessment, opening the door to new diagnostics for environmental toxicology. The mitochondrial stress signature aligns with pathways already being targeted by companies developing senolytics, mitoprotective agents, and anti‑inflammatory therapeutics. The authors wrote that the research provides “a mechanistic foundation for assessing the health risks of plastic pollution and developing therapeutic interventions for environmentally driven biliary disorders.”
The post Microplastics in Human Bile Drive Mitochondrial Dysfunction and Senescence appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors