Uncategorized
Accelerating Organoid Adoption Set to Transform Cancer Drugs
Since the FDA’s April 2025 announcement to phase out required animal testing in favor of human-first preclinical models for monoclonal antibody therapies and other drugs, the adoption of organoids that mimic the structure, function, and cellular complexity of native tissues has been on the rise. These ex vivo systems provide a more physiologically relevant platform for evaluating drug efficacy and toxicity, with the potential to improve clinical outcomes for patients.
The momentum behind this regulatory shift was underscored last October by the first FDA investigational new drug (IND) approval in oncology based exclusively on efficacy data from human vascularized organoids, generated using a platform developed by Qureator.
In cancer research, organoid biobanks that capture diverse genetic backgrounds, ethnicities, ages, and other patient-specific variables are opening new avenues for precision medicine.
Yet Hans Clevers, MD, PhD, renowned stem cell biologist and the “father of organoids,” explains that replacing decades of infrastructure reliant on animal models is not an easy feat.
“In Dutch, we call this koudwatervrees—cold water fear,” said Clevers during his keynote session at GEN’s Spotlight on Organoids, which streamed in March. “During my seven years with Roche, I learned there are thousands of individual technologies in drug development proven over decades. You cannot simply discard those without providing the same level of evidence.”
Today’s organoid companies underscore proof points in the ability to model complex human biology with high fidelity. At the same time, the government’s evolving regulatory stance is serving as a critical catalyst, providing both validation and momentum to accelerate widespread adoption of these platforms.
Organoid intelligence
When asked about the biggest obstacles preventing organoids from reaching their full potential, Alif Saleh, CEO of 28bio, emphasizes that adoption, not technology, is the primary hurdle, particularly in drug discovery, where a staggering 95% failure rate is accepted as the norm.
“The risk of failure is built in all the way to Wall Street,” said Saleh. “If everybody accepts that risk, there’s very little incentive to change.” He states that achieving a large cultural shift for widespread organoid adoption will likely require either regulatory pressure or an organoid-powered commercial success.
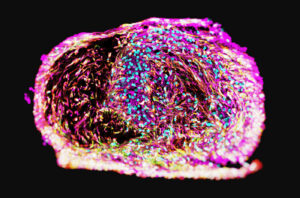
28bio builds human brain organoids at scale that model memory and learning improvement in neurodegenerative disease. The platform grows tissue in the lab programmed for key neurological cell types, including both microglia and oligodendrocytes, by starting from primary or induced pluripotent stem cells (iPSCs).
These brain organoids predict drug efficacy and toxicity in both the central and peripheral nervous systems. In cancer research, the model provides a valuable tool for screening antibody drug conjugate (ADC) neurotoxicity.
Saleh says organoids are “becoming smarter and smarter.” 28bio’s neural interface measures the effects of different compounds on memory and task performance by recording what it calls “organoid intelligence.” The platform trains brain organoids to play simple games such as Pac-Man, providing stimulation that rewards successful behaviors (e.g., “eating” fruit) and discourages failures (e.g., colliding with ghosts). By quantifying learning and performance changes in response to drug exposure, the system provides a functional readout of neurological impact in vitro.
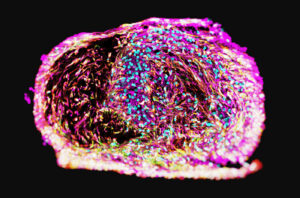
28bio is not the only player in the brain organoid space. BrainStorm Therapeutics is building AI models that develop gene maps of brain diseases, which then identify promising targets for new drugs and clinical biomarkers. The company’s organoid platform tackles both rare brain diseases, such as Rett Syndrome and CDKL5 deficiency disorder, and common neurodegenerative conditions, like Parkinson’s and Alzheimer’s. The platform screens thousands of drug molecules per day on the model’s human brain cells.
“It doesn’t matter in the end, how well designed your drug is,” said Maya Gosztyla, PhD, co-founder and CSO at BrainStorm, at GEN’s Spotlight on Organoids. “When we brought AI into our platform, we tried to take a biology-first approach that trains models on the richest and most accurate datasets directly from human patient-derived brain organoid models.”
BrainStorm is a member of NVIDIA Inception, a program that guides AI startups through the NVIDIA ecosystem of compute technologies. The company’s brain organoids generate single-cell sequencing data to fine-tune foundation models available through NVIDIA’s BioNeMo Framework, including the Geneformer model for gene expression analysis.
Can’t ignore the immune system
“Have you ever bet $3 billion on a single number in roulette? That’s essentially drug discovery,” said Robert DiFazio, PhD, co-founder at Parallel Bio, in response to pharma’s high failure rates. “Pharma needs to recognize that animal testing is a problem.”

Parallel Bio seeks to model the complex immune system by developing replicas of human lymph nodes that demonstrate biological behavior, such as swelling during inflammation and the formation of germinal centers. These transient microstructures are critical to immune response and allow antigen-activated B cells to proliferate and differentiate into high-affinity memory B cells and plasma cells. The company aims to fuel immunotherapies for autoimmune disease and cancer.
Juliana Hilliard, PhD, co-founder at Parallel Bio, states that a dual organoid approach, where systemic immunity is paired with other tissues, such as cancer tumors, provides a more representative model for human disease.
The company’s organoids can capture immune suppression, checkpoint activity, cytokine dynamics, immune cell infiltration, and tumor cell killing factors that are often absent in other preclinical models. By exploring immune-cancer interactions at high resolution, the platform can support patient stratification, informed drug design for defined populations, and prediction of immuno-oncology drug efficacy prior to clinical trials.
“COVID was the first time you saw a rush of pharma companies trying to hire immunologists because they couldn’t ignore the immune system anymore,” said Hilliard. With increasing evidence that organoids can successfully flag drug failures before Phase I clinical trials, she foresees human-first drug discovery as inevitable.
“Eventually, pharma won’t be able to develop drugs in animals,” Hilliard says.
Ludovico Buti, PhD, senior research leader at Charles River Laboratories, affirms that the immune system is critical for modeling complex cancer biology. He emphasizes that cancer organoids have historically focused on the epithelial components of the tumor. Gaps remain when recapitulating the stroma and immune components.
“We have been able to circumvent this challenge by co-culturing organoids with immune cells that come from the same donor, if you’re lucky, or from different donors, but this was always considered a transitory solution,” Buti explained. “We want the resident immune cells already embedded in the organoids. That’s how we now see the technology evolving today.”
Capture the microenvironment
Homing in on cancer, Henriette Lanz, PhD, VP of biology at MIMETAS, affirms that patient-derived tumor organoids (PDOs) have already taken flight in recent years prior to the FDA’s announcements. She explains that initiatives, such as the Organoids Platform within the Oncode Accelerator program in the Netherlands, were launched several years ago to accelerate the development of new cancer therapies for the benefit of patients of all ages. The group is also investing in a large PDO biobank that is accessible to drug developers.
PDOs maintain heterogeneity of the original tumor tissue while capturing inter-patient variability. Yet, these models are limited by the lack of a tumor microenvironment, which is composed of immune cells, vasculature, and supporting cell types, such as fibroblasts.
To address this gap, MIMETAS’ OrganoPlate Graft UniFlow technology creates vascularized co-cultures to support immune-competent tumors-on-chip with unidirectional perfusion. The system simplifies operation by eliminating the requirement for external pumps and supports up to 512 chips in a single setup for large-scale screening. Additionally, MIMETAS has a colon organoid tubular model on the market for toxicity prediction and a kidney organoid tubular model under early access.
Lorna Ewart, PhD, CSO of Emulate, affirms that the publication of the FDA’s April 2025 roadmap signals a move toward using animals as “the exception rather than the norm.” Reduced perceived regulatory risk has led to increased use of organ-on-a-chip technologies that use microfluidic devices to precisely control the cellular microenvironment and model tumor biology.
She says these regulatory changes encourage end users in pharmaceutical companies and beyond to evaluate the decision-quality of the data as a critical next step to incorporate into their regular workflows, with the ultimate goal of impacting patients.
“It is widely appreciated that the earlier a patient is diagnosed and correctly treated, the higher their chances of progression-free or overall survival are likely to be,” said Ewart. “What excites me most about the future of organ-on-a-chip research in cancer is the ability to improve patient care.”
To model tumor biology, Emulate’s platform introduces stromal contents by tuning the extracellular matrix stiffness and composition, as well as recreating essential gradients. The method captures dynamic processes, such as immune cell recruitment and drug exposure, to assist in the testing of therapeutic candidates.

Emulate’s first-generation platform, Zoë CM-2, has been used internally to assess CAR T cell recruitment, as well as externally by researchers at the Ellison Institute and Wyss Institute to model cancer cell extravasation and Barrett’s esophageal cancer, respectively. Additionally, researchers at McGill University Health Center demonstrated that the organ-on-a-chip models could predict a cancer patient’s response to chemotherapy.
Last June, Emulate launched AVA Emulation System, a self-contained, benchtop culture module that incubates and images up to 96 organ-chip samples in a single run. According to an Emulate press release, the platform cuts consumable costs fourfold and in-lab labor by half compared to current generation technologies.
While these human-first models have outperformed animal models, predicting clinical outcomes directly remains a reach. Clevers asserts that organoids are poised to become a key solution for almost all challenges in drug development, a vision that must be coordinated globally to be achieved.
“My dream is that in the next 10 years, we will make this technology a commodity,” he says. “Something where people can just push a button and it works.”
The post Accelerating Organoid Adoption Set to Transform Cancer Drugs appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Opinion: STAT+: Former Geisinger CEO: U.S. health systems must replace huge numbers of people with AI
About 20 years ago, I stepped on stage at one of our Geisinger town halls and looked out upon a sea of people: thousands of full-time employees at an integrated health system charged with the health and well-being of millions of Pennsylvanians.
Only a fraction of the people in that room were clinicians.
That was the first time I fully visualized the problem: We employed more people in our revenue cycle department to process bills and reconcile data than we did doctors. And we weren’t alone. It’s the same story at every health system in America, large and small, and over the past two decades, the ratio has become dramatically more disparate.

About 20 years ago, I stepped on stage at one of our Geisinger town halls and looked out upon a sea of people: thousands of full-time employees at an integrated health system charged with the health and well-being of millions of Pennsylvanians.
Only a fraction of the people in that room were clinicians.
That was the first time I fully visualized the problem: We employed more people in our revenue cycle department to process bills and reconcile data than we did doctors. And we weren’t alone. It’s the same story at every health system in America, large and small, and over the past two decades, the ratio has become dramatically more disparate.
Uncategorized
A star scientist showed that better genetics lessons could reduce racism. It was the death knell for his career
Every year, the Genetics Society of America bestows the Elizabeth W. Jones Award for Excellence in Education, recognizing someone who has helped the public better understand the science of DNA. It’s understood to be a lifetime achievement award; past recipients tend toward retirement age with decades of work behind them and stacks of textbooks to their names.
When this year’s winner, Brian Donovan, was announced at the end of February, many geneticists and science educators found it hard to celebrate the news. Not because he’s undeserving of the honor. Far from it. But because it seemed to confirm what many feared: that Donovan’s incandescent research career was over before it had barely begun.

Every year, the Genetics Society of America bestows the Elizabeth W. Jones Award for Excellence in Education, recognizing someone who has helped the public better understand the science of DNA. It’s understood to be a lifetime achievement award; past recipients tend toward retirement age with decades of work behind them and stacks of textbooks to their names.
When this year’s winner, Brian Donovan, was announced at the end of February, many geneticists and science educators found it hard to celebrate the news. Not because he’s undeserving of the honor. Far from it. But because it seemed to confirm what many feared: that Donovan’s incandescent research career was over before it had barely begun.
Uncategorized
Microplastics in Human Bile Drive Mitochondrial Dysfunction and Senescence
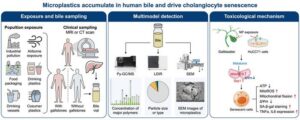
Microplastics have become a defining environmental signature of modern life, turning up in oceans, soil, food, drinking water, and even the air. But their biological fate inside the human body remains far less understood. A new study suggests that these particles may be doing more than simply passing through. Instead, they may be accumulating in one of the body’s most overlooked fluids—bile—and leaving behind measurable cellular damage that could shape future thinking about environmentally driven biliary injury and long‑term health effects. As the authors noted in their abstract, “the long-term accumulation patterns and chronic toxic effects of microplastics within the human biliary system are largely unknown,” underscoring the need for deeper investigation into how these particles behave in the enterohepatic circulation.
Researchers from the Tenth Affiliated Hospital of Southern Medical University (Dongguan People’s Hospital), Sun Yat-sen University, Guilin Medical University, and collaborating institutions reported the findings in Environmental Science and Ecotechnology. Their study, “Microplastics accumulate in human bile and drive cholangiocyte senescence,” provides the first direct evidence that microplastics are not only present in bile but may also contribute to mitochondrial dysfunction and premature aging in cholangiocytes, the epithelial cells that line the bile ducts.
The team collected bile from 14 surgical patients (five without gallstones and nine with gallstones) and used a multimodal analytical approach—pyrolysis–gas chromatography–mass spectrometry, laser direct infrared spectroscopy, and scanning electron microscopy—to characterize the particles. According to the paper, “we show the universal presence of microplastics in human bile,” identifying six polymer types dominated by polyethylene terephthalate and polyethylene, with most particles measuring 20–50 μm. Patients with gallstones carried substantially higher microplastic burdens, raising questions about whether biliary stasis or altered bile composition may influence microplastic retention.

To probe biological effects, the researchers exposed cultured human cholangiocytes to low-dose polystyrene nanoplastics for seven days, simulating chronic exposure. The cells exhibited mitochondrial dysfunction, elevated reactive oxygen species, reduced ATP, Drp1‑mediated mitochondrial fission, and G1 cell‑cycle arrest—hallmarks of senescence. As the authors wrote, chronic exposure “induces mitochondrial dysfunction-associated senescence in cholangiocytes,” suggesting a mechanistic link between environmental microplastics and biliary aging.
One of the most intriguing findings is that melatonin, a widely used antioxidant, partially reversed the mitochondrial and inflammatory damage. While far from a therapeutic recommendation, the result hints at a potential intervention point and gives the study translational relevance.
The work reframes the biliary system as something far more active than a simple transit channel. The data indicate that bile can serve as a reservoir for microplastics and that prolonged exposure may age cholangiocytes by driving mitochondrial dysfunction. The partial rescue with melatonin adds a mechanistic foothold for future intervention, even as the authors caution that broader human studies are essential.
For biotech, the implications are broad. The work highlights bile as a clinically accessible matrix for exposure assessment, opening the door to new diagnostics for environmental toxicology. The mitochondrial stress signature aligns with pathways already being targeted by companies developing senolytics, mitoprotective agents, and anti‑inflammatory therapeutics. The authors wrote that the research provides “a mechanistic foundation for assessing the health risks of plastic pollution and developing therapeutic interventions for environmentally driven biliary disorders.”
The post Microplastics in Human Bile Drive Mitochondrial Dysfunction and Senescence appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors