Uncategorized
New protein drugs could pave way for faster drug development
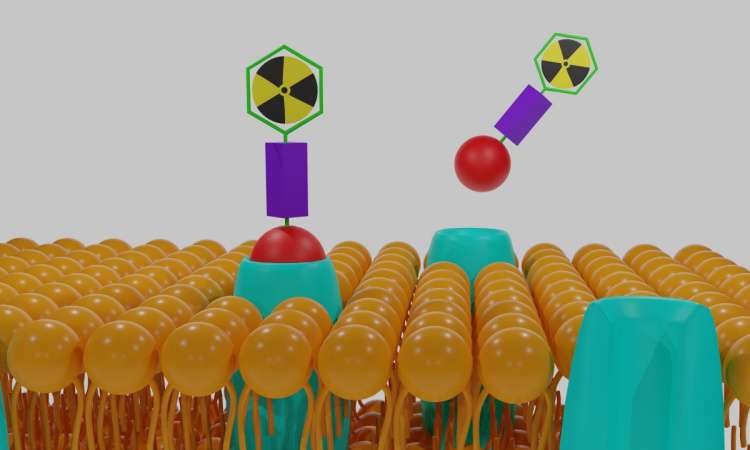

A new class of protein drugs have shown amenability to multiple isotopes, paving the way for faster radiopharmaceutical drug development.
Biotechnology company Molecular Partners announced preclinical data showing its Radio-DARPins are suitable to different isotopes, each specific for a different tumour target.
DARPin (Designed Ankyrin Repeat Protein) therapeutics are a novel class of protein drugs based on natural binding proteins, which have been clinically validated across several therapeutic areas and developed through to the registrational stage.
The key properties of DARPins – intrinsic high affinity and specificity, small size, flexible architecture, and high stability – offer unmatched advantages to drug design, such as multi-specificity, broad target range, and tuneable half-life.
The results of studies in tumour-bearing mice show highly comparable biodistribution profiles for both Radio-DARPin candidates labelled with Lutetium-177 (177Lu) or with Lead-203 (203Pb), with similar uptake and washout rates. Imaging with 177Lu can be indicative of behaviour with the therapeutic isotope Actinium-225 (225Ac), and similarly with 203Pb for 212Pb.
The preclinical data was revealed at the Global Radiopharmaceuticals Development Summit, which took place in Shanghai, China on March 19–20.
“Our recent data confirms that our Radio-DARPin-vector design allows interchangeability of alpha-isotopes, including 212Pb and 225Ac,” said Patrick Amstutz, CEO of Molecular Partners.
“This feature offers us the opportunity and flexibility to evaluate Radio-DARPin candidates in an isotope-agnostic manner and to choose the most suitable therapeutic isotope, as late as with initial clinical data, without having to restart the entire drug discovery and development process – a significant advantage to tailor our candidates to patient needs.”
The post New protein drugs could pave way for faster drug development appeared first on Drug Discovery World (DDW).
Uncategorized
Neurocrine Grows in Endocrinology, Rare Disease with $2.9B Soleno Buyout
Neurocrine Biosciences has agreed to acquire Soleno Therapeutics for $2.9 billion, the companies said, in a deal designed to bolster the buyer’s portfolio of marketed endocrinology and rare disease therapies.
“This transaction will advance Neurocrine’s mission to deliver life-changing treatments while accelerating our revenue growth and portfolio diversification strategy,” Kyle W. Gano, PhD, Neurocrine’s CEO, said in a statement.
The acquisition would bolster Neurocrine’s offerings to include three treatments that have already reached the market:
- Crenessity® (crinecerfont), a treatment of classic congenital adrenal hyperplasia (CAH) due to 21-hydroxylase deficiency that received FDA approval in December 2024
- Ingrezza® (valbenazine), a vesicular monoamine transmitter 2 (VMAT2) drug approved in 2017 as a treatment for tardive dyskinesia and the chorea associated with Huntington’s disease
- Vykat
 XR (diazoxide choline), approved last year as the first and only therapy indicated to treat hyperphagia in patients ages four and older with Prader-Willi syndrome (PWS).
XR (diazoxide choline), approved last year as the first and only therapy indicated to treat hyperphagia in patients ages four and older with Prader-Willi syndrome (PWS).
“Neurocrine is the right strategic partner to expand the reach of Vykat XR in the Prader-Willi syndrome community given their experience in endocrinology and rare disease and their proven ability to execute successful commercial launches,” stated Anish Bhatnagar, MD, Soleno’s chairman and CEO. “We are excited to accelerate Vykat XR’s impact for PWS patients following completion of the transaction by leveraging Neurocrine’s strong commercial capabilities.”
Soleno finished 2025 with $190.4 million in net revenue from sales of Vykat XR—including $91.7 million generated during the fourth quarter, pushing the company to profitability with positive net income of $20.9 million.
‘A little surprising’
Stifel analysts Paul Matteis and James Condulis called the planned acquisition “a little surprising” since Vykat XR is projected to garner approximately $400 million in annual net revenue, he commented in a note reported by Bloomberg News.
Vykat XR won FDA approval in March 2025. From then through December 31, 859 active patients were prescribed the drug by 630 unique prescribers (136 of them in Q4), while the company received 1,250 patient start forms (207 in Q4).
Neurocrine expects Vykat XR’s numbers to improve in coming years, since the drug is positioned as a foundational first-line therapy for PWS and is supported by a patent portfolio that is expected to protect the drug’s exclusivity into the mid-2040s.
Vykat XR would join Neurocrine’s marketed portfolio which includes Ingrezza and Crenessity. Ingrazza racked up blockbuster net revenue numbers of $2.51 billion up 9% year-over-year (including $657.5 million during Q4, up 7% from the year-ago quarter). Neurocrine has credited double-digit prescription volume growth in total prescriptions and new (first-time) prescriptions, partially offset by a lower net price that the company called new “formulary access investments” designed to support long-term growth.
Crenessity generated $301.2 million in net product sales last year for Neurocrine, including $135.3 million in the fourth quarter, reflecting 2,048 total new patient enrollment start forms, 431 of them in Q4 2025.
Neurocrine reasons that the three drugs will deliver sustained revenue growth for the combined company through the end of the decade.
Also for Neurocrine, a buyout of Soleno presents a “more sensible way into metabolic disease” than by developing its own pipeline candidates, which are in preclinical phases, and risking competitive and regulatory challenges, BMO Capital Markets analyst Evan Seigerman observed in a research note reported by Reuters.
Neurocrine has disclosed plans to begin Phase I studies this year for two preclinical obesity candidates: NBIP-‘2118, a CRF2 agonist; and ‘NBIP-‘1968, a combination of ‘2118 and the company’s own GIP (glucose-dependent insulinotropic polypeptide)/ GLP-1 (glucagon-like peptide-1) preferring triple agonist, which Neurocrine calls “light” on glucagon activity.
News of a potential buyout of Soleno by Neurocrine was first reported Sunday by the Financial Times.
Soleno investors signaled approval of the buyout Monday by sending shares to $52.25, up 32% from Thursday’s close of $39.49 (Markets were closed Friday for Good Friday). However, Neurocrine’s investors weren’t as supportive of the deal as that company’s shares barely budged, closing at $132.48, up 0.67% from $131.60 on Thursday.
Second thoughts?
A potential reason: Neurocrine investors may have second thoughts about a deal that would add to its pipeline Vykat XR, whose prescribing label includes warnings and precautions about past reports of hyperglycemia and fluid retention/edema, as Sumant Kulkarni, a senior analyst covering biotechnology with Canaccord Genuity, commented in a research note.
“We believe NBIX would have to articulate its plans very well for investors to display enthusiasm from the get-go,” Kulkarni wrote.
Yet two things could work in Neurocrine’s favor, Kulkarni added: The company’s solid track record of commercialization as seen with Ingrezza and Crenessity, and the prospect of adding to the portfolio Vykat XR given its approval for a rare form of obesity.
San Diego-based Neurocrine reported approximately 2,000 employees as of December 31, 2025, with plans during the first quarter to complete the expansion of sales teams for Ingrezza and Crenessity “to maximize our commercial momentum.” Soleno is based in Redwood City, CA, and reported a workforce of 182 full-time employees as of the end of 2025.
At $53 per share cash, the purchase price represents a premium of about 34% above Soleno’s closing share price Thursday, and a premium of 51% to Soleno’s 30-day volume-weighted average price (VWAP).
The boards of both Neurocrine and Soleno have approved the transaction, which is expected to close within 90 days subject to satisfying customary closing conditions that include receipt of regulatory approvals.
Neurocrine will acquire Soleno by launching a tender offer for that company’s outstanding shares. Following a successful completion of the tender offer, a wholly owned subsidiary of Neurocrine will merge with Soleno, and the outstanding Soleno shares not tendered in the offer will be converted into the right to receive the same $53 per share in cash paid in the tender offer.
Consummation of the tender offer is subject to the tender of at least a majority of the outstanding shares of Soleno, the expiration or termination of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, and other customary conditions.
Neurocine said it will fund its acquisition of Soleno using a “modest amount” of pre-payable debt plus cash on hand. Neurocrine reported $1.48 billion in cash, cash equivalents, and marketable securities as of December 31, 2025—up 37.5% from $1.076 billion a year earlier.
The post Neurocrine Grows in Endocrinology, Rare Disease with $2.9B Soleno Buyout appeared first on GEN – Genetic Engineering and Biotechnology News.
Uncategorized
Did Eli Lilly just strike another gold mine?
Did Eli Lilly just strike another gold mine?
Eli Lilly may not have discovered orexins, but the pharma’s latest acquisition marks increasing momentum for a drug class that’s maturing after years of fine-tuning.
The big question on the minds of Wall Street is … Read More
Uncategorized
White House adds AbbVie’s Humira to TrumpRx alongside cheaper biosimilars
White House adds AbbVie’s Humira to TrumpRx alongside cheaper biosimilars
The White House this week began offering a $950-per-month version of AbbVie’s megablockbuster Humira on TrumpRx, even as the drug discount website also offers two Humira biosimilars from Pfizer and Amgen that both cost less … Read More
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors