Uncategorized
STAT+: New obesity tool aims to predict risk of 18 serious complications
Body mass index has its limitations, but for now it’s the metric medicine often defaults to when predicting weight-related health problems. A new tool promises to better define who’s at risk for obesity complications, based on measures that include BMI but also family history, diet, current illness, and socioeconomic factors culled from medical records.
One aim of the research is to better understand who’s a candidate for an obesity drug, often prescribed based on BMI alone or BMI in combination with another disease. Over time, GLP-1 medications, whose initial target was type 2 diabetes, have revealed their power to ease cardiovascular disease, kidney disease, liver disease, sleep apnea, and osteoarthritis, in addition to promoting significant weight loss. But discerning who’s the best fit for the costly, lifelong treatment has been uncertain.
“We really wanted to have an integrated model that enables us to look at not one, but 18 different obesity-relevant complications,” Claudia Langenberg, co-author of a study about the new model published Thursday in Nature Medicine, said in a media briefing Tuesday. She is director and professor of medicine and population health at Precision Healthcare University Research Institute of Queen Mary University of London.
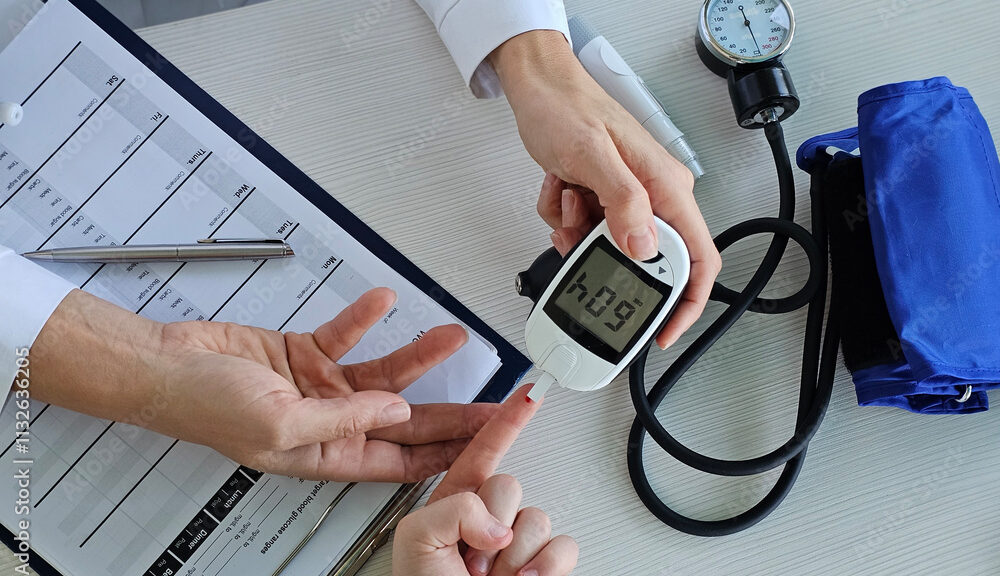
Body mass index has its limitations, but for now it’s the metric medicine often defaults to when predicting weight-related health problems. A new tool promises to better define who’s at risk for obesity complications, based on measures that include BMI but also family history, diet, current illness, and socioeconomic factors culled from medical records.
One aim of the research is to better understand who’s a candidate for an obesity drug, often prescribed based on BMI alone or BMI in combination with another disease. Over time, GLP-1 medications, whose initial target was type 2 diabetes, have revealed their power to ease cardiovascular disease, kidney disease, liver disease, sleep apnea, and osteoarthritis, in addition to promoting significant weight loss. But discerning who’s the best fit for the costly, lifelong treatment has been uncertain.
“We really wanted to have an integrated model that enables us to look at not one, but 18 different obesity-relevant complications,” Claudia Langenberg, co-author of a study about the new model published Thursday in Nature Medicine, said in a media briefing Tuesday. She is director and professor of medicine and population health at Precision Healthcare University Research Institute of Queen Mary University of London.
Uncategorized
CVS’ healthcare delivery chief eyes potential in clinical AI
CVS Health has been testing AI tools that deliver healthcare, though it sees too many barriers to using them more widely for now.
CVS’ President of Health Care Delivery Sree Chaguturu told Endpoints News at …
Uncategorized
Lilly clocks 56% revenue increase in Q1 but declining prices stunt growth
Eli Lilly’s $19.8 billion revenue for the first quarter could have been higher if not for declining prices for key medicines like Zepbound, Mounjaro and Taltz.
Uncategorized
Bioprocessing Applications Laboratory Opened in Korea by Ecolab Life Sciences
Officials at Ecolab Life Sciences report that the company is expanding its bioprocessing business with the launch of a new bioprocessing applications lab (BPAL) in Dongtan, Korea. They say the goal is to provide biopharmaceutical manufacturers across Asia with better local access to downstream process development support.
The site is Ecolab’s first bioprocessing facility in Asia and joins an established applications network in the U.S. and U.K.
BPAL Korea supports process development from early-stage resin screening through studies designed to replicate commercial manufacturing conditions, according to Jenny Tan, vice president and general manager, Ecolab Life Sciences APAC and India. On-site scientists work alongside customers across Asia to help optimize chromatography steps, improve yield and productivity, and accelerate regulatory pathways, with the aim of reducing the need to ship resins and reference materials overseas for development work, she continues.
Asia has become one of the world’s most active biopharmaceutical manufacturing regions, with Korea, China, Japan, India, and Singapore all home to growing pipelines in biosimilars and monoclonal antibody processes that scalable downstream purification. With local technical support now in place, manufacturers across the region can shorten development cycles and maintain consistency with global operations while working to tight regulatory and cost targets, continues Tan.
“Biopharmaceutical manufacturers across Asia are under increasing pressure to scale with speed while meeting demanding regulatory and performance expectations,” she explains. “BPAL Korea strengthens our ability to work side by side with customers, bringing local expertise together with Ecolab’s global, integrated bioprocessing network.”
By combining local scientific support with Ecolab’s innovative Purolite resin portfolio, Ecolab’s new BPAL was created to help enable manufacturers to address process challenges earlier, reduce development risk, and advance programs with greater confidence as they prepare for scaleup, says Tan.
resin portfolio, Ecolab’s new BPAL was created to help enable manufacturers to address process challenges earlier, reduce development risk, and advance programs with greater confidence as they prepare for scaleup, says Tan.
The post Bioprocessing Applications Laboratory Opened in Korea by Ecolab Life Sciences appeared first on GEN – Genetic Engineering and Biotechnology News.
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars



