Uncategorized
STAT+: AIDS group sues Trump administration over undisclosed agreement with Gilead
An AIDS activist group filed a lawsuit against the Trump administration for failing to disclose a research and development agreement that was at the heart of a settlement between the U.S. government and Gilead Sciences over patents for HIV prevention.
The settlement resolved a contentious lawsuit that was filed six years ago by the previous Trump administration after the Centers for Disease Control and Prevention maintained that Gilead infringed its patent rights. The agency had helped fund academic research that later formed the basis for two Gilead HIV pills, Truvada and Descovy.
The administration had alleged that Gilead ignored the contributions by CDC scientists, exaggerated its own role in developing HIV prevention drugs, and refused to sign a licensing agreement despite “multiple attempts” at reaching a deal after unfairly reaping hundreds of millions of dollars from research funded by taxpayers.
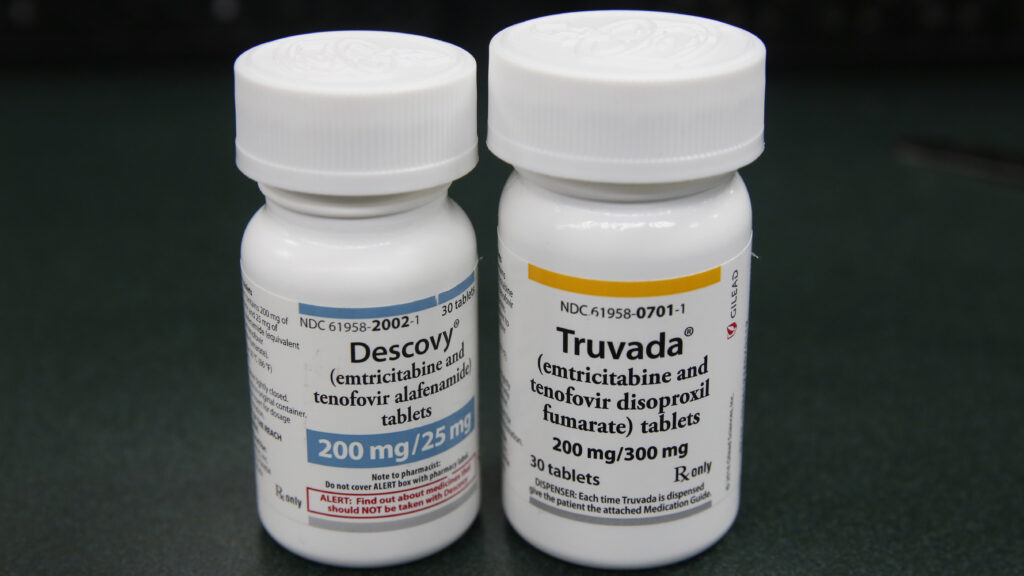
An AIDS activist group filed a lawsuit against the Trump administration for failing to disclose a research and development agreement that was at the heart of a settlement between the U.S. government and Gilead Sciences over patents for HIV prevention.
The settlement resolved a contentious lawsuit that was filed six years ago by the previous Trump administration after the Centers for Disease Control and Prevention maintained that Gilead infringed its patent rights. The agency had helped fund academic research that later formed the basis for two Gilead HIV pills, Truvada and Descovy.
The administration had alleged that Gilead ignored the contributions by CDC scientists, exaggerated its own role in developing HIV prevention drugs, and refused to sign a licensing agreement despite “multiple attempts” at reaching a deal after unfairly reaping hundreds of millions of dollars from research funded by taxpayers.
Uncategorized
STAT+: Johnson & Johnson advances IBD therapy, despite trial miss
A closely watched therapy developed by Johnson & Johnson failed to show a statistically meaningful improvement for patients with inflammatory bowel disease. But the company plans to advance the drug into late-stage testing, focusing on a growing subgroup of patients.
On Tuesday, trial investigators presented the results of Johnson & Johnson’s DUET study, testing how well combining the drugs Tremfya and Simponi would stop the immune system from mistakenly attacking healthy tissues in the digestive tract. It’s a follow-up to a 2022 clinical trial that showed a significant benefit to patients, nearly doubling the rates of disease remission and spurring several companies to start developing combination approaches for IBD.
Johnson & Johnson tested its combined therapy, dubbed JNJ-4804, in two Phase 2b clinical trials hitting both major forms of inflammatory bowel disease — ulcerative colitis and Crohn’s disease. In each trial, the combination therapy performed better than the individual drugs, but did not meet the primary endpoint of clinical remission.

A closely watched therapy developed by Johnson & Johnson failed to show a statistically meaningful improvement for patients with inflammatory bowel disease. But the company plans to advance the drug into late-stage testing, focusing on a growing subgroup of patients.
On Tuesday, trial investigators presented the results of Johnson & Johnson’s DUET study, testing how well combining the drugs Tremfya and Simponi would stop the immune system from mistakenly attacking healthy tissues in the digestive tract. It’s a follow-up to a 2022 clinical trial that showed a significant benefit to patients, nearly doubling the rates of disease remission and spurring several companies to start developing combination approaches for IBD.
Johnson & Johnson tested its combined therapy, dubbed JNJ-4804, in two Phase 2b clinical trials hitting both major forms of inflammatory bowel disease — ulcerative colitis and Crohn’s disease. In each trial, the combination therapy performed better than the individual drugs, but did not meet the primary endpoint of clinical remission.
Uncategorized
Sertraline manufacturer recalls antidepressant batch after UK packaging mix-up
A subsidiary of Indian drugmaker Hetero Group mistakenly packaged two types of antidepressants in packs of sertraline, potentially posing risks to patients.
Uncategorized
Vertex drops mRNA cystic fibrosis program over ‘tolerability’ issues
Vertex Pharmaceuticals said it has dropped development of an mRNA-based cystic fibrosis therapy, after facing challenges delivering the genetic medicine similar to those that have troubled other parts of the field.
The Boston-based company
-

 Uncategorized9 years ago
Uncategorized9 years agoThese ’90s fashion trends are making a comeback in 2017
-

 Contributors9 years ago
Contributors9 years agoThe final 6 ‘Game of Thrones’ episodes might feel like a full season
-

 Uncategorized9 years ago
Uncategorized9 years agoAccording to Dior Couture, this taboo fashion accessory is back
-

 Uncategorized9 years ago
Uncategorized9 years agoUber and Lyft are finally available in all of New York State
-

 Uncategorized9 years ago
Uncategorized9 years agoPhillies’ Aaron Altherr makes mind-boggling barehanded play
-

 Uncategorized9 years ago
Uncategorized9 years agoThe old and New Edition cast comes together to perform
-

 Uncategorized9 years ago
Uncategorized9 years agoSteph Curry finally got the contract he deserves from the Warriors
-

 Uncategorized9 years ago
Uncategorized9 years agoDisney’s live-action Aladdin finally finds its stars





